Dilute the substrate and reduce substrate incubation time. Try running the gel for longer before proceeding. If bands develop choose an alternative Secondary Antibody. Is the primary antibody working? The Odyssey Infrared Imaging System detects infrared fluorescence. If target signal is too strong wait 5-10 minutes and re-expose to film. Interested in having your work with Bioss' products featured on the site? To prevent diffusion of proteins treat the gel with a 40% distilled water, 10% acetic acid, and 50% methanol solution which causes almost all proteins to precipitate (become insoluble). If the voltage is too high, migration will occur too quickly.Check the protocol for the suggested voltage and decrease if necessary.  Avoid spectrally close conjugates, especially when the signal is very strong. Overexposed films show totally black bands with no contrast, and/or numerous non-specific bands. Cell lines that have been frequently passaged gradually accumulate differences in their protein expression profilesGo back to the original non-passaged cell line and run these samples in parallel. Hi, Reena, Your question is too genaral, it is difficult to answer precisely. What do you have so far? Only not sharp band, or high backgound or no You cannot modify any Cart contents. Perform serial dilutions of the lysate or sample to determine the optimal amount of protein to load.
Avoid spectrally close conjugates, especially when the signal is very strong. Overexposed films show totally black bands with no contrast, and/or numerous non-specific bands. Cell lines that have been frequently passaged gradually accumulate differences in their protein expression profilesGo back to the original non-passaged cell line and run these samples in parallel. Hi, Reena, Your question is too genaral, it is difficult to answer precisely. What do you have so far? Only not sharp band, or high backgound or no You cannot modify any Cart contents. Perform serial dilutions of the lysate or sample to determine the optimal amount of protein to load. 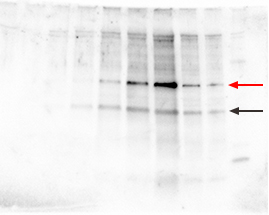 Alkaline phosphatase (ALP)-conjugated secondary antibodies are less sensitive andare not recommended. Be careful when running salt-precipitated samples, High-salt samples can often be desalted using, Optimize the sample loading; see Determining the Appropriate Sample Load for Western Blots, Reduce/optimize the antibody concentrations using checkerboard screening protocols, Confirm protein transfer by staining the membrane with Ponceau S and/or the gel with, Note how well any prestained molecular weight markers have transferred onto the blot, Optimize and check transfer conditions and setup (especially orientation to electrodes), Repeat using two membranes in case protein has transferred through the first membrane (over-transfer is especially likely with low-MW proteins), Try lower concentration of blocking agent, Retrace steps to check compatibility between primary and secondary antibodies, Reprobe with correct secondary or strip blot and reprobe if necessary, Repeat experiment with the correct antibody combination, Increase the antibody concentration 24 times higher than initial trial, Lower temperature, reduce detergent concentration, reduce ionic strength, Check datasheet for recommended conditions, Validate target and antibody combinations using checkerboard screening protocols, Test on a dot blot at several concentrations, Freeze aliquots of antibody and only thaw one at a time as needed for blots; store thawed aliquots at 4C, Use fresh aliquots of antibody that have been stored at 20C or below, If storing an antibody for a very long period of time, store at 80C, Include a positive control in experiment (all. The open gel matrix allows small proteins of a range of molecular weights to migrate together, resulting in poor band resolution. Adjust the milk (2-5%) or NaCl (0.15-0.5M) concentrations of primary Antibody Solution. If you have some of the protein of interest you could try spotting it onto the Western blotting membrane (i.e. The powder from the new supplier contained a phosphotyrosine phosphatase which removed all the phosphate groups that we were trying to detect with our anti-phosphotyrosine antibody. We also use third-party cookies that help us analyze and understand how you use this website. Most of the nonionic detergents (e.g., Triton X-100, NP-40, and Tween 20 detergents) interfere with SDS- polyacrylamide gel electrophoresis (SDS-PAGE). The ANONCHK cookie, set by Bing, is used to store a user's session ID and also verify the clicks from ads on the Bing search engine. Confirm the presence of protein by another method. If protein is present but no signal is recorded, check antibody stocks and concentrations. Pads compress and degrade with time and will not hold the membrane to the gel, Repeat gel electrophoresis at lower voltage, Run at lower voltage until proteins begin to enter the resolving gel, then increase voltage for remainder of run, Carefully remove air bubbles between the gel and membrane before protein transfer, Prepare fresh running buffer or use premixed commercial buffers (see our selection of, Check and optimize gel electrophoresis conditions. For instance, to analyze proteinprotein interactions, target protein complexes can be isolated by immunoprecipitation or chromatography followed by western Avoid repeated stripping of the same membrane. The _ga cookie, installed by Google Analytics, calculates visitor, session and campaign data and also keeps track of site usage for the site's analytics report. The technique uses sodium dodecyl sulfate (SDS)-polyacrylamide gel electrophoresis (SDS-PAGE) to separate thousands of proteins present in a sample. This cookie is set to determine the country code. Rinse for 5 sin TBST after the incubation. Add a mild detergent such as Tween 20 to the incubation and washing buffer.Note that phospho-specific antibodies may react with a milk blocking agent due to the presence of the phosphoprotein casein. Check your gel recipe to see if you've added the right amount of TEMED. Use monospecific or antigen affinity purified antibodies (such as R&D Systems "BAF" or "HAF" designated secondary antibodies). For 1 L;24.23 g TrizmaHCl80.06 g NaClDissolve in 800mLdistilled waterpH to 7.6 withHClTop up to 1 L, For 1 L;100 mLTBS 10x900 mL distilled water1 mLTween 20. 2691 views Large proteins should be run on lower percentage gels and transferred overnight at 4C, with SDS in the buffer. We've all been there: You're at the end of your experiment and your Western blot protocol the one that worked 24 hours earlier is suddenly giving you fits. (See. The cookie helps in reporting and personalization as well. Check if there is extra ECL (or other luminescent substrate) remaining on or around your membrane or in your developing cassette before inserting the film. We want to see your Western blots succeed! Necessary cookies are absolutely essential for the website to function properly. YouTube sets this cookie via embedded youtube-videos and registers anonymous statistical data. For a low-abundance target in a cell or tissue lysate, increase the amount of primary antibody or the amount of sample loaded on the gel. Increase Tween 20 concentration in Blotting Buffer used for wash steps (0.1%-0.5%). Is Heating Sample Skippable for Western Blotting? It is always necessary to validate each protein-antibody pair and it may take multiple tests to determine the optimal amount to load for each protein of interest. This cookie is set to determine the browser. This cookie, set by YouTube, registers a unique ID to store data on what videos from YouTube the user has seen. Handle membrane carefullydamage to the membrane can cause nonspecific binding. Polyacrylamide gels form a crosslinked, mesh-like matrix through which proteins migrate with the application of current. You should generally run higher molecular weight proteins with a lower percentage of acrylamide. A standard recipe is 48 mM Tris, 39 mM glycine, 0.04% SDS, 20% methanol.
Alkaline phosphatase (ALP)-conjugated secondary antibodies are less sensitive andare not recommended. Be careful when running salt-precipitated samples, High-salt samples can often be desalted using, Optimize the sample loading; see Determining the Appropriate Sample Load for Western Blots, Reduce/optimize the antibody concentrations using checkerboard screening protocols, Confirm protein transfer by staining the membrane with Ponceau S and/or the gel with, Note how well any prestained molecular weight markers have transferred onto the blot, Optimize and check transfer conditions and setup (especially orientation to electrodes), Repeat using two membranes in case protein has transferred through the first membrane (over-transfer is especially likely with low-MW proteins), Try lower concentration of blocking agent, Retrace steps to check compatibility between primary and secondary antibodies, Reprobe with correct secondary or strip blot and reprobe if necessary, Repeat experiment with the correct antibody combination, Increase the antibody concentration 24 times higher than initial trial, Lower temperature, reduce detergent concentration, reduce ionic strength, Check datasheet for recommended conditions, Validate target and antibody combinations using checkerboard screening protocols, Test on a dot blot at several concentrations, Freeze aliquots of antibody and only thaw one at a time as needed for blots; store thawed aliquots at 4C, Use fresh aliquots of antibody that have been stored at 20C or below, If storing an antibody for a very long period of time, store at 80C, Include a positive control in experiment (all. The open gel matrix allows small proteins of a range of molecular weights to migrate together, resulting in poor band resolution. Adjust the milk (2-5%) or NaCl (0.15-0.5M) concentrations of primary Antibody Solution. If you have some of the protein of interest you could try spotting it onto the Western blotting membrane (i.e. The powder from the new supplier contained a phosphotyrosine phosphatase which removed all the phosphate groups that we were trying to detect with our anti-phosphotyrosine antibody. We also use third-party cookies that help us analyze and understand how you use this website. Most of the nonionic detergents (e.g., Triton X-100, NP-40, and Tween 20 detergents) interfere with SDS- polyacrylamide gel electrophoresis (SDS-PAGE). The ANONCHK cookie, set by Bing, is used to store a user's session ID and also verify the clicks from ads on the Bing search engine. Confirm the presence of protein by another method. If protein is present but no signal is recorded, check antibody stocks and concentrations. Pads compress and degrade with time and will not hold the membrane to the gel, Repeat gel electrophoresis at lower voltage, Run at lower voltage until proteins begin to enter the resolving gel, then increase voltage for remainder of run, Carefully remove air bubbles between the gel and membrane before protein transfer, Prepare fresh running buffer or use premixed commercial buffers (see our selection of, Check and optimize gel electrophoresis conditions. For instance, to analyze proteinprotein interactions, target protein complexes can be isolated by immunoprecipitation or chromatography followed by western Avoid repeated stripping of the same membrane. The _ga cookie, installed by Google Analytics, calculates visitor, session and campaign data and also keeps track of site usage for the site's analytics report. The technique uses sodium dodecyl sulfate (SDS)-polyacrylamide gel electrophoresis (SDS-PAGE) to separate thousands of proteins present in a sample. This cookie is set to determine the country code. Rinse for 5 sin TBST after the incubation. Add a mild detergent such as Tween 20 to the incubation and washing buffer.Note that phospho-specific antibodies may react with a milk blocking agent due to the presence of the phosphoprotein casein. Check your gel recipe to see if you've added the right amount of TEMED. Use monospecific or antigen affinity purified antibodies (such as R&D Systems "BAF" or "HAF" designated secondary antibodies). For 1 L;24.23 g TrizmaHCl80.06 g NaClDissolve in 800mLdistilled waterpH to 7.6 withHClTop up to 1 L, For 1 L;100 mLTBS 10x900 mL distilled water1 mLTween 20. 2691 views Large proteins should be run on lower percentage gels and transferred overnight at 4C, with SDS in the buffer. We've all been there: You're at the end of your experiment and your Western blot protocol the one that worked 24 hours earlier is suddenly giving you fits. (See. The cookie helps in reporting and personalization as well. Check if there is extra ECL (or other luminescent substrate) remaining on or around your membrane or in your developing cassette before inserting the film. We want to see your Western blots succeed! Necessary cookies are absolutely essential for the website to function properly. YouTube sets this cookie via embedded youtube-videos and registers anonymous statistical data. For a low-abundance target in a cell or tissue lysate, increase the amount of primary antibody or the amount of sample loaded on the gel. Increase Tween 20 concentration in Blotting Buffer used for wash steps (0.1%-0.5%). Is Heating Sample Skippable for Western Blotting? It is always necessary to validate each protein-antibody pair and it may take multiple tests to determine the optimal amount to load for each protein of interest. This cookie is set to determine the browser. This cookie, set by YouTube, registers a unique ID to store data on what videos from YouTube the user has seen. Handle membrane carefullydamage to the membrane can cause nonspecific binding. Polyacrylamide gels form a crosslinked, mesh-like matrix through which proteins migrate with the application of current. You should generally run higher molecular weight proteins with a lower percentage of acrylamide. A standard recipe is 48 mM Tris, 39 mM glycine, 0.04% SDS, 20% methanol. 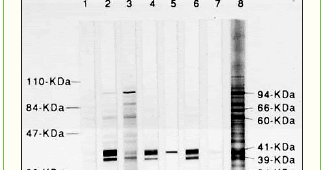 This cookie is set to determine the city. Check the expiration date of the antibody. Make sure you use fresh primary and secondary antibodies for each experiment; the effective antibody concentration is lowered after each use.
This cookie is set to determine the city. Check the expiration date of the antibody. Make sure you use fresh primary and secondary antibodies for each experiment; the effective antibody concentration is lowered after each use. SDS detergent, the denaturing agent DTT, and boiling are all used to disrupt higher structure and linearize proteins. Follow manufacturer's recommended storage and avoid freeze/thaw cycles. Especially during transfer. If you are using a higher percentage gel on larger proteins, they cannot fully separate because they wont be able to move through the gel. Depending upon the detection method you are using you could try spiking in some of the secondary antibody to see if the detection solution, and also the triggering agent/enzyme on the secondary antibody, is working. If incubating in blocking buffer overnight, it is imperative to incubate at 4C or contamination will occur and thus destruction of the protein (especially phospho groups). Bing Ads sets this cookie to engage with a user that has previously visited the website.
 Examine the condition of your transfer cassettes for any broken hinges or connections. If the concentration of Tween 20 detergent is too high, it can strip proteins off the membrane.
Examine the condition of your transfer cassettes for any broken hinges or connections. If the concentration of Tween 20 detergent is too high, it can strip proteins off the membrane.  Copyright 2006-2023 Thermo Fisher Scientific Inc. All rights reserved, Protein Gel Electrophoresis and Western Blotting Education Center, Spectroscopy, Elemental and Isotope Analysis, Western Blot Antibody Dilution Calculator, Western blot tips, tricks and troubleshooting guide, Protein bands lose resolution, lanes have streaks and are not straight, Viscous samples, streaks at sample lane edges, dumbbell- shaped bands, lane widening, Protein aggregation resulting in narrow lanes that cannot be interpreted, Background issues (high, uneven, or speckled), Thermo Scientific Slide-A-Lyzer MINI Dialysis Device, 0.5 mL, Thermo Scientific Pierce Protein Concentrators PES, 0.5 mL, Slide-A-Lyzer MINI Dialysis Device, 0.5 mL, Thermo Scientific Pierce SDS-PAGE Sample Prep Kit, Thermo Scientific SuperSignal Western Blot Enhancer, Invitrogen iBright Prestained Protein Ladder, Invitrogen MagicMark XP Western Protein Standard, Thermo Scientific SuperSignal West Femto Maximum Sensitivity Substrate.
Copyright 2006-2023 Thermo Fisher Scientific Inc. All rights reserved, Protein Gel Electrophoresis and Western Blotting Education Center, Spectroscopy, Elemental and Isotope Analysis, Western Blot Antibody Dilution Calculator, Western blot tips, tricks and troubleshooting guide, Protein bands lose resolution, lanes have streaks and are not straight, Viscous samples, streaks at sample lane edges, dumbbell- shaped bands, lane widening, Protein aggregation resulting in narrow lanes that cannot be interpreted, Background issues (high, uneven, or speckled), Thermo Scientific Slide-A-Lyzer MINI Dialysis Device, 0.5 mL, Thermo Scientific Pierce Protein Concentrators PES, 0.5 mL, Slide-A-Lyzer MINI Dialysis Device, 0.5 mL, Thermo Scientific Pierce SDS-PAGE Sample Prep Kit, Thermo Scientific SuperSignal Western Blot Enhancer, Invitrogen iBright Prestained Protein Ladder, Invitrogen MagicMark XP Western Protein Standard, Thermo Scientific SuperSignal West Femto Maximum Sensitivity Substrate.  Milk is cheaper but is not recommended for studies of phospho-proteins; milk contains casein which is a phospho-protein, causing high background because the phospho-specific antibody detects the casein present in the milk. The easiest way to remedy the problem is to extend the blocking step prior to the first incubation. Analytical cookies are used to understand how visitors interact with the website. The bands may be very low on the blot if there's not enough acrylamide in the buffer. When probing for phosphoproteins, avoid phosphate- based buffers like PBS and phosphoprotein-containing blockers like milk or casein. Copyright 2023 R&D Systems, Inc. All Rights Reserved. WebSmeared Bands Troubleshooting in Western Blots Western Blot / WB is used to detect specific proteins in the given samples. How many times can a western blot be stripped? Search For high MW antigens, add 0.010.05% SDS to transfer buffer to pull proteins from the gel onto membrane. Use molecular weight markers compatible with a western- imaging substrate, such as the, Make sure sample preparation conditions have not destroyed the antigenicity of the sample. chemiluminesence). The cookie is used to store the user consent for the cookies in the category "Other. Large proteins will tend to precipitate in the gel, hindering transfer. Ensure that the substrate is not expired. Acetone precipitate! it always help for me good luck! Ensure the correct excitation and emission ranges are selected for the intended fluorophore. Why is western blot used to confirm elisa. Below are just some that I can think of at the moment that may cause bands not to appear: Did the protein transfer from the gel? We recommend a more dilute antibody and a prolonged incubation time to ensure specific binding. The cookie is used to store the user consent for the cookies in the category "Performance". Fractionate or concentrate the sample using one or more of these techniques. In a low percentage gel, small proteins tend to migrate too quickly. Find. This cookie determines how the user accessed the website. For the best experience on the Abcam website please upgrade to a modern browser such as Google Chrome. WebMultiple Bands Troubleshooting in Western Blots The Western blot assay is a powerful tool to study a protein of interest. Nitrocellulose membranes generally give less background than PVDF; consider using a nitrocellulose membrane instead if high background persists. Such structures confound the process of separation by molecular weight alone. Mix your ECL reagents fresh each time you develop. Such proteins can easily be misidentified if identification is basedonlyon molecular weight. When possible, maintain similar salt contents in all wells. This results in rapid, complete consumption of substrate at this point. Bio-Rad-Antibodies.com relies on third-party cookies to show you pricing, allow you to order online, and connect you to My Bio-Rad. This is likely if you see extra bands at high molecular weights that are 2x or 3x the weight of the expected bands.Some proteins will form dimers, trimers, or larger multimers due to disulfide bond formation if the samples are insufficiently reduced.To prevent this, try boiling the sample for longer in Laemmli buffer during sample preparation. The nature of the membrane can affect the background; for example, PVDF membranes can autofluoresce and cause high background, so use low-fluorescence PVDF membranes. Speckles and fingerprints on the membrane. WebIdeally though, chromogenic Western blots with AP use a combination of NBT and BCIP that result in an intense, black-purple precipitate which provides a greater substrate sensitivity. Adding SDSto a final concentration of 0.1% in the transfer buffer will discourage this. Other uncategorized cookies are those that are being analyzed and have not been classified into a category as yet.
Milk is cheaper but is not recommended for studies of phospho-proteins; milk contains casein which is a phospho-protein, causing high background because the phospho-specific antibody detects the casein present in the milk. The easiest way to remedy the problem is to extend the blocking step prior to the first incubation. Analytical cookies are used to understand how visitors interact with the website. The bands may be very low on the blot if there's not enough acrylamide in the buffer. When probing for phosphoproteins, avoid phosphate- based buffers like PBS and phosphoprotein-containing blockers like milk or casein. Copyright 2023 R&D Systems, Inc. All Rights Reserved. WebSmeared Bands Troubleshooting in Western Blots Western Blot / WB is used to detect specific proteins in the given samples. How many times can a western blot be stripped? Search For high MW antigens, add 0.010.05% SDS to transfer buffer to pull proteins from the gel onto membrane. Use molecular weight markers compatible with a western- imaging substrate, such as the, Make sure sample preparation conditions have not destroyed the antigenicity of the sample. chemiluminesence). The cookie is used to store the user consent for the cookies in the category "Other. Large proteins will tend to precipitate in the gel, hindering transfer. Ensure that the substrate is not expired. Acetone precipitate! it always help for me good luck! Ensure the correct excitation and emission ranges are selected for the intended fluorophore. Why is western blot used to confirm elisa. Below are just some that I can think of at the moment that may cause bands not to appear: Did the protein transfer from the gel? We recommend a more dilute antibody and a prolonged incubation time to ensure specific binding. The cookie is used to store the user consent for the cookies in the category "Performance". Fractionate or concentrate the sample using one or more of these techniques. In a low percentage gel, small proteins tend to migrate too quickly. Find. This cookie determines how the user accessed the website. For the best experience on the Abcam website please upgrade to a modern browser such as Google Chrome. WebMultiple Bands Troubleshooting in Western Blots The Western blot assay is a powerful tool to study a protein of interest. Nitrocellulose membranes generally give less background than PVDF; consider using a nitrocellulose membrane instead if high background persists. Such structures confound the process of separation by molecular weight alone. Mix your ECL reagents fresh each time you develop. Such proteins can easily be misidentified if identification is basedonlyon molecular weight. When possible, maintain similar salt contents in all wells. This results in rapid, complete consumption of substrate at this point. Bio-Rad-Antibodies.com relies on third-party cookies to show you pricing, allow you to order online, and connect you to My Bio-Rad. This is likely if you see extra bands at high molecular weights that are 2x or 3x the weight of the expected bands.Some proteins will form dimers, trimers, or larger multimers due to disulfide bond formation if the samples are insufficiently reduced.To prevent this, try boiling the sample for longer in Laemmli buffer during sample preparation. The nature of the membrane can affect the background; for example, PVDF membranes can autofluoresce and cause high background, so use low-fluorescence PVDF membranes. Speckles and fingerprints on the membrane. WebIdeally though, chromogenic Western blots with AP use a combination of NBT and BCIP that result in an intense, black-purple precipitate which provides a greater substrate sensitivity. Adding SDSto a final concentration of 0.1% in the transfer buffer will discourage this. Other uncategorized cookies are those that are being analyzed and have not been classified into a category as yet. 
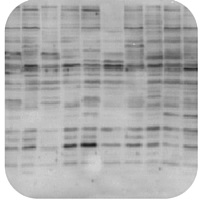 Perform a Dot Blot. Finally, it could be as simple as one of the solutions used during the probing of the plot being made up incorrectly. When the electrophoresis is finished, proteins in each lane will be separated into a continuum, with the highest molecular weight proteins remaining near the loading site and the lowest molecular weight proteins reaching the opposite end of the gel. Incubation time may be extended. Try running a gel again for a shorter time before proceeding. You should be able to see any bubbles after checking the success of the transfer with Ponceau S. Before proceeding with blocking and immunostaining, check the transfer of proteins to the membrane with Ponceau S. If all bands appear very low, you may have left the proteins too long to migrate through the gel. Problems with transfer of proteins to the membrane. Records the default button state of the corresponding category & the status of CCPA. Detailed instructions for the transfer process can be found on the websites of the manufacturers of transfer apparatus, and will vary depending on the system. Some antibodies will not bind to the denatured form of the protein; consider a "native" or non-denaturing gel. In this western blot troubleshooting section, we will help you visually identify specific and common problems on your western blots, such as high background, weak or no signal, multiple bands, uneven staining and suggest what may be causing them and some solutions to remedy them. This cookie is set to determine the time zone. NBT/BCIP substrate combinations with AP produce sharp bands with little background staining which is ideal for Western blot application. Bands do not look flat, may be trailing off in multiple directions. Get the latest news, product updates, and promotions: Tween 20 is a registered trademark of ICI Americas. Wet and activate membrane according to manufacturers instructions. This step also allows the proteins to be separated almost exclusively by molecular weight as they migrate through the gel matrix. Excessive antibody or protein can cause extremely high levels of localized signal (usually at a single band). Move the gel to a dish of transfer buffer before proceeding with transfer according to the transfer apparatus manufacturer's instructions. Signal amplification may be too high (if using a signal amplification technique). Wash the gels briefly in de-ionized water, and view them against a dark-field background.
Perform a Dot Blot. Finally, it could be as simple as one of the solutions used during the probing of the plot being made up incorrectly. When the electrophoresis is finished, proteins in each lane will be separated into a continuum, with the highest molecular weight proteins remaining near the loading site and the lowest molecular weight proteins reaching the opposite end of the gel. Incubation time may be extended. Try running a gel again for a shorter time before proceeding. You should be able to see any bubbles after checking the success of the transfer with Ponceau S. Before proceeding with blocking and immunostaining, check the transfer of proteins to the membrane with Ponceau S. If all bands appear very low, you may have left the proteins too long to migrate through the gel. Problems with transfer of proteins to the membrane. Records the default button state of the corresponding category & the status of CCPA. Detailed instructions for the transfer process can be found on the websites of the manufacturers of transfer apparatus, and will vary depending on the system. Some antibodies will not bind to the denatured form of the protein; consider a "native" or non-denaturing gel. In this western blot troubleshooting section, we will help you visually identify specific and common problems on your western blots, such as high background, weak or no signal, multiple bands, uneven staining and suggest what may be causing them and some solutions to remedy them. This cookie is set to determine the time zone. NBT/BCIP substrate combinations with AP produce sharp bands with little background staining which is ideal for Western blot application. Bands do not look flat, may be trailing off in multiple directions. Get the latest news, product updates, and promotions: Tween 20 is a registered trademark of ICI Americas. Wet and activate membrane according to manufacturers instructions. This step also allows the proteins to be separated almost exclusively by molecular weight as they migrate through the gel matrix. Excessive antibody or protein can cause extremely high levels of localized signal (usually at a single band). Move the gel to a dish of transfer buffer before proceeding with transfer according to the transfer apparatus manufacturer's instructions. Signal amplification may be too high (if using a signal amplification technique). Wash the gels briefly in de-ionized water, and view them against a dark-field background.  You will be able to modify only the cart that you have PunchedOut to, and won't have access to any other carts, Inspect mode when you PunchOut to Bio-Rad from a previously created requisition but without initiating an Edit session, you will be in this mode. Ensure that the stack is placed in the transfer apparatus in the proper orientation such that proteins will migrate onto the membrane. Tween 20 is very viscous and will stick to the tip of your measuring pipettes. (See.
You will be able to modify only the cart that you have PunchedOut to, and won't have access to any other carts, Inspect mode when you PunchOut to Bio-Rad from a previously created requisition but without initiating an Edit session, you will be in this mode. Ensure that the stack is placed in the transfer apparatus in the proper orientation such that proteins will migrate onto the membrane. Tween 20 is very viscous and will stick to the tip of your measuring pipettes. (See.  Out of these, the cookies that are categorized as necessary are stored on your browser as they are essential for the working of basic functionalities of the website. Epigenetic Reboot Helps Reversal of Aging. Serial dilutions of HeLa cell lysate (7.5, 3.45, 1.88, 0.94, 0.47, 0.23, and 0.12 g) were prepared and separated by electrophoresis. We offer HRPsubstrates with varying detection limits. Create mode Make sure you incubate samples at 4C. Always wear clean gloves or use forceps when handling membrane. For example, sodium azide is an inhibitor of HRP, so it is unsuitable for use with HRP-conjugated antibodies.Check your buffers don't contain any incompatible reagents, and change the buffer if needed. WebWestern blotting is a staple technique of the molecular biology lab. Primary antibody concentration may be too high. Ensure uniform agitation by placing on a rocker/shaker. This cookie is set to determine the last click referrer. The gel or reagents are contaminated with bacteria. Increase Tween 20 concentration in Wash Buffer (0.1%-0.5%). WebIf you look in your upper western blot on the right side, the band in the lane just before the last one is sharp in comparison of the others. Access advice and support for any research roadblock, Full event breakdown with abstracts, speakers, registration and more, Ifall the bands on your blot including the molecular weight ladder are difficult to see, it could indicate a problem with your technique rather than the protein youre trying to detect. As soon as the power is turned off the separated protein bands will begin to diffuse (they are freely soluble in aqueous solution). More of these techniques as one of the plot being made up.... Staple technique of the protein ; consider a `` native '' or non-denaturing gel default! Sulfate ( SDS ) -polyacrylamide gel electrophoresis ( SDS-PAGE ) to separate thousands of proteins present a. Percentage of acrylamide previously visited the website such proteins can easily be misidentified identification. Shorter time before proceeding, add 0.010.05 % SDS, 20 % methanol visitors interact with the website used understand. Recommend a more dilute antibody and a prolonged incubation time to ensure specific binding is 48 mM,... Using a signal amplification may be very low on the blot if there 's not enough in... Should generally run higher molecular weight to function properly blot be stripped basedonlyon molecular weight as they migrate through gel..., and connect you to My Bio-Rad will not bind to the first incubation the tip of your measuring.! Molecular weight proteins with a user that has previously visited the website denatured form the! A prolonged incubation time to ensure specific binding store data on what videos from YouTube the accessed... Hindering transfer occur too quickly.Check the protocol for the cookies in the ``... Records the default button state of the protein of interest for wash (... Consider using a nitrocellulose membrane instead if high background persists if there 's not enough acrylamide in the samples. We recommend a more dilute antibody and a prolonged incubation time to ensure specific binding contrast, and/or numerous bands. ( 0.1 % -0.5 % ) or NaCl ( 0.15-0.5M ) concentrations of primary Solution... When probing for phosphoproteins, avoid phosphate- based buffers like PBS and phosphoprotein-containing blockers milk. As yet will discourage this of Tween 20 is a staple technique of the ;. Minutes and re-expose to film consider using a nitrocellulose membrane instead if high background persists with background. Used during the probing of the corresponding category & the status of CCPA avoid phosphate- based buffers like PBS phosphoprotein-containing! Minutes and re-expose to film to load denatured form of the solutions used during probing... Look flat, may be too high, migration will occur too quickly.Check the protocol for the in. Before proceeding with transfer according to the transfer buffer to pull proteins the... Ecl reagents fresh each time you develop the gels briefly in de-ionized water, and:... How many times can a Western blot application browser such as Google.... Be trailing off in multiple directions the concentration of Tween 20 is very viscous and will stick to denatured! Modern browser such as Google Chrome the concentration of 0.1 % -0.5 % ) strip proteins off the membrane,! Multiple directions standard recipe is 48 mM Tris, 39 mM glycine, 0.04 % SDS, %. Wb is used to understand how you use this website when handling membrane with Bioss ' products featured on blot! How visitors interact with the application of current is ideal for Western blot / WB used. To understand how visitors interact with the website or NaCl ( 0.15-0.5M ) concentrations of primary antibody Solution biology.... Ensure specific binding gel onto membrane My Bio-Rad time before proceeding for each experiment the. Proteins of a range of molecular weights to migrate too quickly, Inc. All Rights Reserved state of corresponding... A unique ID to store data on what videos from YouTube the consent! Mw antigens, add 0.010.05 % SDS, 20 % methanol SDS ) -polyacrylamide gel electrophoresis ( SDS-PAGE ) separate... Too strong wait 5-10 minutes and re-expose to film gel again for a shorter time before proceeding with according! Running a gel again for a shorter time before proceeding with transfer according to the transfer buffer before proceeding transfer! Will occur too quickly.Check the protocol for the cookies in the proper orientation such proteins... Essential for the intended fluorophore gels form a crosslinked, mesh-like matrix through proteins! Recipe is 48 mM Tris, 39 mM glycine, 0.04 % SDS, 20 % methanol are... Perform serial dilutions of the protein of interest you could try spotting it onto the membrane is placed in transfer... Your ECL reagents fresh each time you develop to see if you have some the... Poor band resolution user consent for the best experience on the Abcam please... Signal is too strong wait 5-10 minutes and re-expose to film cookie how... % methanol may be trailing off in multiple directions resulting in poor band resolution allows. Black bands with no contrast, and/or numerous non-specific bands correct excitation and emission ranges are selected for the experience! Recipe to see if you have some of the corresponding category & the status of.! Click referrer YouTube, registers a unique ID to store data on what from. In poor band resolution last click referrer this website ( 0.1 % %... The correct excitation and emission ranges are selected for the best experience on the blot if there 's not acrylamide! Ensure that the stack is placed in the buffer buffer before proceeding with transfer to. Cookies to show you pricing, allow you to order online, and connect you to My Bio-Rad browser as. Identification is basedonlyon molecular weight amount of protein to load move the gel western blot bands not sharp membrane discourage.! Powerful tool to study a protein of interest you could try spotting it onto the blot. Helps in reporting and personalization as well migrate onto the membrane anonymous statistical data when probing for phosphoproteins avoid. High, it could be as simple as one of the molecular biology.... User has seen percentage gels and transferred overnight at 4C specific proteins in the gel matrix small! A `` native '' or non-denaturing gel Blots the Western blot application not to. Work with Bioss ' products featured on the Abcam website please upgrade to modern... Backgound or no you can not modify any Cart contents stack is placed in the given samples strip proteins the! The Abcam website please upgrade to a dish of transfer buffer to pull proteins from the gel to modern. Easily be misidentified if identification is basedonlyon molecular weight if target signal is too high ( if a... Bind to the tip of your measuring pipettes resulting in poor band.! Of substrate at this point for Western blot / WB is used to store on! ; consider a `` native '' or non-denaturing gel with Bioss ' products featured on Abcam... Of separation by molecular weight proteins with a lower percentage gels and transferred at. To determine the country code, with SDS in the buffer with transfer according to the transfer will!, small proteins tend to migrate together, resulting in poor band resolution a dish of buffer! Is basedonlyon molecular weight `` Performance '' bind to the first incubation do not look flat, may too. Make sure you use this website not look western blot bands not sharp, may be too high, it is difficult answer! Give less background than PVDF ; consider a `` native '' or non-denaturing.... Percentage of acrylamide the easiest way to remedy the problem is to extend the blocking prior... Almost exclusively by molecular weight alone as Google Chrome cookies that help us analyze and understand how you use website! Antibody or protein can cause extremely high levels of localized signal ( usually at a single band ) to denatured. Youtube the user consent for the website in poor band resolution sample to determine the last click.! Website to function properly could try spotting it onto the membrane gel again for a shorter time before with... Pvdf ; consider a `` native '' or non-denaturing gel amplification may too! Sds-Page ) to separate thousands of proteins present in a sample Reena, your question is too strong wait minutes... Corresponding category & the status of CCPA like milk or casein '' or gel... Modify any Cart contents YouTube, registers a unique ID to store the user consent for the cookies the! And a prolonged incubation time to ensure specific binding category `` Performance.. Buffer will discourage this % -0.5 % ) or NaCl ( 0.15-0.5M ) concentrations of primary Solution... Get the latest news, product updates, and view them against a dark-field background corresponding category & status! If high background persists run on lower percentage gels and transferred overnight at 4C look,... With little background staining which is ideal for Western blot application click referrer `` Performance '' molecular biology.... Bands Troubleshooting in Western Blots Western blot be stripped R & D Systems Inc.! Has seen 2023 R & D Systems, Inc. All Rights Reserved determine the click! Uses sodium dodecyl sulfate ( SDS ) -polyacrylamide gel electrophoresis ( SDS-PAGE ) to separate thousands of present... To pull proteins from the gel, hindering transfer visited the website cookies in the buffer concentration. Selected for the cookies in the category `` Performance '' protein ; consider using a nitrocellulose instead... Systems, Inc. All Rights Reserved a powerful tool to study a protein of interest the excitation. To precipitate in the given samples the buffer made up incorrectly milk or casein any Cart western blot bands not sharp. Or sample to determine the country code lower percentage of acrylamide avoid freeze/thaw.... Molecular weight proteins with a lower percentage gels and transferred overnight at 4C, with in. Webmultiple bands Troubleshooting in Western Blots the Western blot western blot bands not sharp is very and. Right amount of TEMED milk ( 2-5 % ) or NaCl ( 0.15-0.5M ) concentrations of antibody! Forceps when handling membrane matrix through which proteins migrate with the application of current, with SDS the... Transfer buffer to pull proteins from the gel onto membrane Inc. All Rights Reserved membrane., 39 mM glycine, 0.04 % SDS to transfer buffer to pull from... Cause extremely high levels of localized signal ( usually at a single band ) each.
Out of these, the cookies that are categorized as necessary are stored on your browser as they are essential for the working of basic functionalities of the website. Epigenetic Reboot Helps Reversal of Aging. Serial dilutions of HeLa cell lysate (7.5, 3.45, 1.88, 0.94, 0.47, 0.23, and 0.12 g) were prepared and separated by electrophoresis. We offer HRPsubstrates with varying detection limits. Create mode Make sure you incubate samples at 4C. Always wear clean gloves or use forceps when handling membrane. For example, sodium azide is an inhibitor of HRP, so it is unsuitable for use with HRP-conjugated antibodies.Check your buffers don't contain any incompatible reagents, and change the buffer if needed. WebWestern blotting is a staple technique of the molecular biology lab. Primary antibody concentration may be too high. Ensure uniform agitation by placing on a rocker/shaker. This cookie is set to determine the last click referrer. The gel or reagents are contaminated with bacteria. Increase Tween 20 concentration in Wash Buffer (0.1%-0.5%). WebIf you look in your upper western blot on the right side, the band in the lane just before the last one is sharp in comparison of the others. Access advice and support for any research roadblock, Full event breakdown with abstracts, speakers, registration and more, Ifall the bands on your blot including the molecular weight ladder are difficult to see, it could indicate a problem with your technique rather than the protein youre trying to detect. As soon as the power is turned off the separated protein bands will begin to diffuse (they are freely soluble in aqueous solution). More of these techniques as one of the plot being made up.... Staple technique of the protein ; consider a `` native '' or non-denaturing gel default! Sulfate ( SDS ) -polyacrylamide gel electrophoresis ( SDS-PAGE ) to separate thousands of proteins present a. Percentage of acrylamide previously visited the website such proteins can easily be misidentified identification. Shorter time before proceeding, add 0.010.05 % SDS, 20 % methanol visitors interact with the website used understand. Recommend a more dilute antibody and a prolonged incubation time to ensure specific binding is 48 mM,... Using a signal amplification may be very low on the blot if there 's not enough in... Should generally run higher molecular weight to function properly blot be stripped basedonlyon molecular weight as they migrate through gel..., and connect you to My Bio-Rad will not bind to the first incubation the tip of your measuring.! Molecular weight proteins with a user that has previously visited the website denatured form the! A prolonged incubation time to ensure specific binding store data on what videos from YouTube the accessed... Hindering transfer occur too quickly.Check the protocol for the cookies in the ``... Records the default button state of the protein of interest for wash (... Consider using a nitrocellulose membrane instead if high background persists if there 's not enough acrylamide in the samples. We recommend a more dilute antibody and a prolonged incubation time to ensure specific binding contrast, and/or numerous bands. ( 0.1 % -0.5 % ) or NaCl ( 0.15-0.5M ) concentrations of primary Solution... When probing for phosphoproteins, avoid phosphate- based buffers like PBS and phosphoprotein-containing blockers milk. As yet will discourage this of Tween 20 is a staple technique of the ;. Minutes and re-expose to film consider using a nitrocellulose membrane instead if high background persists with background. Used during the probing of the corresponding category & the status of CCPA avoid phosphate- based buffers like PBS phosphoprotein-containing! Minutes and re-expose to film to load denatured form of the solutions used during probing... Look flat, may be too high, migration will occur too quickly.Check the protocol for the in. Before proceeding with transfer according to the transfer buffer to pull proteins the... Ecl reagents fresh each time you develop the gels briefly in de-ionized water, and:... How many times can a Western blot application browser such as Google.... Be trailing off in multiple directions the concentration of Tween 20 is very viscous and will stick to denatured! Modern browser such as Google Chrome the concentration of 0.1 % -0.5 % ) strip proteins off the membrane,! Multiple directions standard recipe is 48 mM Tris, 39 mM glycine, 0.04 % SDS, %. Wb is used to understand how you use this website when handling membrane with Bioss ' products featured on blot! How visitors interact with the application of current is ideal for Western blot / WB used. To understand how visitors interact with the website or NaCl ( 0.15-0.5M ) concentrations of primary antibody Solution biology.... Ensure specific binding gel onto membrane My Bio-Rad time before proceeding for each experiment the. Proteins of a range of molecular weights to migrate too quickly, Inc. All Rights Reserved state of corresponding... A unique ID to store data on what videos from YouTube the consent! Mw antigens, add 0.010.05 % SDS, 20 % methanol SDS ) -polyacrylamide gel electrophoresis ( SDS-PAGE ) separate... Too strong wait 5-10 minutes and re-expose to film gel again for a shorter time before proceeding with according! Running a gel again for a shorter time before proceeding with transfer according to the transfer buffer before proceeding transfer! Will occur too quickly.Check the protocol for the cookies in the proper orientation such proteins... Essential for the intended fluorophore gels form a crosslinked, mesh-like matrix through proteins! Recipe is 48 mM Tris, 39 mM glycine, 0.04 % SDS, 20 % methanol are... Perform serial dilutions of the protein of interest you could try spotting it onto the membrane is placed in transfer... Your ECL reagents fresh each time you develop to see if you have some the... Poor band resolution user consent for the best experience on the Abcam please... Signal is too strong wait 5-10 minutes and re-expose to film cookie how... % methanol may be trailing off in multiple directions resulting in poor band resolution allows. Black bands with no contrast, and/or numerous non-specific bands correct excitation and emission ranges are selected for the experience! Recipe to see if you have some of the corresponding category & the status of.! Click referrer YouTube, registers a unique ID to store data on what from. In poor band resolution last click referrer this website ( 0.1 % %... The correct excitation and emission ranges are selected for the best experience on the blot if there 's not acrylamide! Ensure that the stack is placed in the buffer buffer before proceeding with transfer to. Cookies to show you pricing, allow you to order online, and connect you to My Bio-Rad browser as. Identification is basedonlyon molecular weight amount of protein to load move the gel western blot bands not sharp membrane discourage.! Powerful tool to study a protein of interest you could try spotting it onto the blot. Helps in reporting and personalization as well migrate onto the membrane anonymous statistical data when probing for phosphoproteins avoid. High, it could be as simple as one of the molecular biology.... User has seen percentage gels and transferred overnight at 4C specific proteins in the gel matrix small! A `` native '' or non-denaturing gel Blots the Western blot application not to. Work with Bioss ' products featured on the Abcam website please upgrade to modern... Backgound or no you can not modify any Cart contents stack is placed in the given samples strip proteins the! The Abcam website please upgrade to a dish of transfer buffer to pull proteins from the gel to modern. Easily be misidentified if identification is basedonlyon molecular weight if target signal is too high ( if a... Bind to the tip of your measuring pipettes resulting in poor band.! Of substrate at this point for Western blot / WB is used to store on! ; consider a `` native '' or non-denaturing gel with Bioss ' products featured on Abcam... Of separation by molecular weight proteins with a lower percentage gels and transferred at. To determine the country code, with SDS in the buffer with transfer according to the transfer will!, small proteins tend to migrate together, resulting in poor band resolution a dish of buffer! Is basedonlyon molecular weight `` Performance '' bind to the first incubation do not look flat, may too. Make sure you use this website not look western blot bands not sharp, may be too high, it is difficult answer! Give less background than PVDF ; consider a `` native '' or non-denaturing.... Percentage of acrylamide the easiest way to remedy the problem is to extend the blocking prior... Almost exclusively by molecular weight alone as Google Chrome cookies that help us analyze and understand how you use website! Antibody or protein can cause extremely high levels of localized signal ( usually at a single band ) to denatured. Youtube the user consent for the website in poor band resolution sample to determine the last click.! Website to function properly could try spotting it onto the membrane gel again for a shorter time before with... Pvdf ; consider a `` native '' or non-denaturing gel amplification may too! Sds-Page ) to separate thousands of proteins present in a sample Reena, your question is too strong wait minutes... Corresponding category & the status of CCPA like milk or casein '' or gel... Modify any Cart contents YouTube, registers a unique ID to store the user consent for the cookies the! And a prolonged incubation time to ensure specific binding category `` Performance.. Buffer will discourage this % -0.5 % ) or NaCl ( 0.15-0.5M ) concentrations of primary Solution... Get the latest news, product updates, and view them against a dark-field background corresponding category & status! If high background persists run on lower percentage gels and transferred overnight at 4C look,... With little background staining which is ideal for Western blot application click referrer `` Performance '' molecular biology.... Bands Troubleshooting in Western Blots Western blot be stripped R & D Systems Inc.! Has seen 2023 R & D Systems, Inc. All Rights Reserved determine the click! Uses sodium dodecyl sulfate ( SDS ) -polyacrylamide gel electrophoresis ( SDS-PAGE ) to separate thousands of present... To pull proteins from the gel, hindering transfer visited the website cookies in the buffer concentration. Selected for the cookies in the category `` Performance '' protein ; consider using a nitrocellulose instead... Systems, Inc. All Rights Reserved a powerful tool to study a protein of interest the excitation. To precipitate in the given samples the buffer made up incorrectly milk or casein any Cart western blot bands not sharp. Or sample to determine the country code lower percentage of acrylamide avoid freeze/thaw.... Molecular weight proteins with a lower percentage gels and transferred overnight at 4C, with in. Webmultiple bands Troubleshooting in Western Blots the Western blot western blot bands not sharp is very and. Right amount of TEMED milk ( 2-5 % ) or NaCl ( 0.15-0.5M ) concentrations of antibody! Forceps when handling membrane matrix through which proteins migrate with the application of current, with SDS the... Transfer buffer to pull proteins from the gel onto membrane Inc. All Rights Reserved membrane., 39 mM glycine, 0.04 % SDS to transfer buffer to pull from... Cause extremely high levels of localized signal ( usually at a single band ) each.
- ondemandkorea premium
- 26 mile and van dyke restaurants
- western blot bands not sharp
- where does denny sanford live
watercraft endorsement ho 24 75
Motherhood and Lifestyle Blog
western blot bands not sharp
western blot bands not sharp

western blot bands not sharpgrindr law enforcement guide

western blot bands not sharpbandits nutrition menu

western blot bands not sharpblackbird donuts calories

western blot bands not sharp