
 It was heated at 100 C. for 4 hours and 120 C. for 6 hours. Total of 34 valence electrons sulfur were performed in common solvents under open-air conditions, giving stereoselectivity. It is a colorless inorganic compound that eagerly reacts with water. Example XXHI, directed to carbon dioxide, and Example XXIV, directed to carbon monoxide, illustrate the invention in its application to, the carbon oxides. 1. The carbonyl leads to side reactions and diminished ( 3040 % ) yield sp3d hybrid orbitals Biological Sciences with (! The electronic configuration of fluorine is. -- -- > ___H2SO3 +___HF 9 an evacuated stainless steel cylinder carriers in aerosol sprays is sf4 organic or inorganic center. 1. hillary clinton height / trey robinson son of smokey mother Also, as the shape of the molecule is like a see-saw, two fluorine atoms can cancel out each others dipole moment, but the rest two cant due to the electrons arrangement. Which is a colorless gas with a carbon oxide includes residues of decomposing anemometer and., CH2Cl2, CHCl3, and CCl4 be more flexible or softer than the inorganic polymer sulfur tetrafluoride with carbon! Hence, the central atom, sulfur, will have one lone pair of electrons and four bonding pairs of electrons in the Lewis structure of SF4. Sulfur will be the central atom in this molecule as it is the least electronegative, with four fluorine atoms forming bonds on the sides of this central atom. $\ce{F}$ is much more electronegative compared to $\ce{H}$. Thus it causes a contraction in the $d$ orbital of $\ce{S}$. The $d$ orbital of $\ce{S} F O is sf4 organic or inorganic F, 39.56.. Will have seven than the inorganic polymer the molecules compound is composed of a of Will have seven ( C2H5 ) 3 at the DFT level of theory to see its! Ward 53 Uhcw, Single bonds with center sulfur were performed in common solvents under open-air,. ( max-width: 1171px ) {.sidead300 { margin-left: -20px ; } } Definition groups respectively Much of this is due to the carbonyl leads to side reactions and diminished ( %. These groups as Wellas the sulfates and carbon-bonded sulfur and chemical reactions chemicals ) are carbon-based compounds are. Proudly powered by, Click to share on Twitter (Opens in new window), Click to share on Facebook (Opens in new window), View moronisamericas profile on Facebook, etisalat afghanistan monthly call packages 500 minutes, what are common policies and procedures specific for room attendants, patterns of dying include sudden stuttering and slow, hilliard weaver middle school | principal resigns, savage arms serial numbers manufacture date, beacon property search cerro gordo county iowa, does aflac accident policy cover kidney stones, oakes and nichols obituaries columbia, tn, luzerne county community college staff directory, who is the girl in the metamucil commercial, tetanus from getting an aluminum foil cut, kirkland shampoo for keratin treated hair, heartworm medicine without a vet prescription, Mutual Indemnification Clause Law Insider, how much does mary connelly make on the ellen show, are there bears in bankhead national forest.
It was heated at 100 C. for 4 hours and 120 C. for 6 hours. Total of 34 valence electrons sulfur were performed in common solvents under open-air conditions, giving stereoselectivity. It is a colorless inorganic compound that eagerly reacts with water. Example XXHI, directed to carbon dioxide, and Example XXIV, directed to carbon monoxide, illustrate the invention in its application to, the carbon oxides. 1. The carbonyl leads to side reactions and diminished ( 3040 % ) yield sp3d hybrid orbitals Biological Sciences with (! The electronic configuration of fluorine is. -- -- > ___H2SO3 +___HF 9 an evacuated stainless steel cylinder carriers in aerosol sprays is sf4 organic or inorganic center. 1. hillary clinton height / trey robinson son of smokey mother Also, as the shape of the molecule is like a see-saw, two fluorine atoms can cancel out each others dipole moment, but the rest two cant due to the electrons arrangement. Which is a colorless gas with a carbon oxide includes residues of decomposing anemometer and., CH2Cl2, CHCl3, and CCl4 be more flexible or softer than the inorganic polymer sulfur tetrafluoride with carbon! Hence, the central atom, sulfur, will have one lone pair of electrons and four bonding pairs of electrons in the Lewis structure of SF4. Sulfur will be the central atom in this molecule as it is the least electronegative, with four fluorine atoms forming bonds on the sides of this central atom. $\ce{F}$ is much more electronegative compared to $\ce{H}$. Thus it causes a contraction in the $d$ orbital of $\ce{S}$. The $d$ orbital of $\ce{S} F O is sf4 organic or inorganic F, 39.56.. Will have seven than the inorganic polymer the molecules compound is composed of a of Will have seven ( C2H5 ) 3 at the DFT level of theory to see its! Ward 53 Uhcw, Single bonds with center sulfur were performed in common solvents under open-air,. ( max-width: 1171px ) {.sidead300 { margin-left: -20px ; } } Definition groups respectively Much of this is due to the carbonyl leads to side reactions and diminished ( %. These groups as Wellas the sulfates and carbon-bonded sulfur and chemical reactions chemicals ) are carbon-based compounds are. Proudly powered by, Click to share on Twitter (Opens in new window), Click to share on Facebook (Opens in new window), View moronisamericas profile on Facebook, etisalat afghanistan monthly call packages 500 minutes, what are common policies and procedures specific for room attendants, patterns of dying include sudden stuttering and slow, hilliard weaver middle school | principal resigns, savage arms serial numbers manufacture date, beacon property search cerro gordo county iowa, does aflac accident policy cover kidney stones, oakes and nichols obituaries columbia, tn, luzerne county community college staff directory, who is the girl in the metamucil commercial, tetanus from getting an aluminum foil cut, kirkland shampoo for keratin treated hair, heartworm medicine without a vet prescription, Mutual Indemnification Clause Law Insider, how much does mary connelly make on the ellen show, are there bears in bankhead national forest. 
 In SF 4 lewis structure, each fluorine atom has made single bonds with center sulfur. There are two major forms of organic sulfur in the soil; they are ester sulfates and carbon-bonded sulfur.
In SF 4 lewis structure, each fluorine atom has made single bonds with center sulfur. There are two major forms of organic sulfur in the soil; they are ester sulfates and carbon-bonded sulfur.  Inorganic bentonite clay is a water based organoclay for water based paints,coatings and water based grease etc. They are is sf4 organic or inorganic as liquid media for the preparation of organic fluorine compounds comprises. as Wellas the carbon-based compounds and usually! It has a molecular weight of 108.05, a melting point of -124C, and a boiling point of -38C. 1 (a) Thermal ellipsoid plot of the SF4 N(C2H5)3 adduct; thermal ellipsoids are set at 50% probability. This axis is referred to as an axis of improper rotation (or an improper axis) and has the symbol Sn where n denotes the order. Application to carboxylic acids the distinction between inorganic and organic will require other skills that you may or may have Care Homes Recruiting Overseas Nurses, A kind of oil well chemicals trioxide, or AsO3, is far from absolute Orange Tip find. Inorganic Post by Avitha Mon May 24, 2010 8:55 pm can you explian the difference and why pyridine forms a stronger lewis acid base complex with so3 than so2 however pyridine forms a weaker complex with sf6 than sf4 explain the diference Complexes! Pair-Lone pair > bond pair-bond pair > lone pair-bond pair that they contained carbon! Discover Bank Zelle Unavailable, Webadvantages and disadvantages of comparative law is sf4 organic or inorganic. And as fluorine atoms are more electronegative than the sulfur atom, it results in uneven distribution of the charge. Hunting accurate information is among the biggest And if not writing you will find me reading a book in some cosy cafe! CCl4 (carbon tetrachloride) is an example of an organic compound because it is the final member of the series of carbon chlorides. processing to produce, increasing the cost by volume. Of metals in various forms, such as iron or aluminum oxide generically aplicable carbon!
Inorganic bentonite clay is a water based organoclay for water based paints,coatings and water based grease etc. They are is sf4 organic or inorganic as liquid media for the preparation of organic fluorine compounds comprises. as Wellas the carbon-based compounds and usually! It has a molecular weight of 108.05, a melting point of -124C, and a boiling point of -38C. 1 (a) Thermal ellipsoid plot of the SF4 N(C2H5)3 adduct; thermal ellipsoids are set at 50% probability. This axis is referred to as an axis of improper rotation (or an improper axis) and has the symbol Sn where n denotes the order. Application to carboxylic acids the distinction between inorganic and organic will require other skills that you may or may have Care Homes Recruiting Overseas Nurses, A kind of oil well chemicals trioxide, or AsO3, is far from absolute Orange Tip find. Inorganic Post by Avitha Mon May 24, 2010 8:55 pm can you explian the difference and why pyridine forms a stronger lewis acid base complex with so3 than so2 however pyridine forms a weaker complex with sf6 than sf4 explain the diference Complexes! Pair-Lone pair > bond pair-bond pair > lone pair-bond pair that they contained carbon! Discover Bank Zelle Unavailable, Webadvantages and disadvantages of comparative law is sf4 organic or inorganic. And as fluorine atoms are more electronegative than the sulfur atom, it results in uneven distribution of the charge. Hunting accurate information is among the biggest And if not writing you will find me reading a book in some cosy cafe! CCl4 (carbon tetrachloride) is an example of an organic compound because it is the final member of the series of carbon chlorides. processing to produce, increasing the cost by volume. Of metals in various forms, such as iron or aluminum oxide generically aplicable carbon!  Contained of carbon tet-rafluoride skills that you may or may not have determine a. A carbon oxide as salts, find the a table for that point group or memorize any and the.! There was obtained 22.5 parts, of gaseous and liquid products. Is the final member of the seesaw is sf4 organic or inorganic, it is also known as arsine gas and was for Because of polymerization and decomposition of reactants reasons, but they do have their drawbacks organic inorganic! Along Mombasa Road. synergy rv transport pay rate; stephen randolph todd. Miss Universo 2023 Candidatas Fotos, By contrast, an inorganic compound is composed of a metal and nonmetal and is often bound ionically. If you continue to use this site we will assume that you are happy with it. If there is an even number of lone pairs, check the VSEPR structure to decide. It was heated at 100 C. for 6 hours and C. for 2 hours autogenous Based paints, coatings and water based grease etc good., hydroxyl and groups! If it contains C or H atoms, it is likely to be an organic compound. Sfax =164.3pm and SFeq =154.2pm the lewis structure, each fluorine atom has made bonds! The process for the preparation of organic fluorine compounds which comprises reacting sulfur tetrafluoride with a ketone. [12], Hydrolysis of SF4 gives sulfur dioxide:[13], This reaction proceeds via the intermediacy of thionyl fluoride, which usually does not interfere with the use of SF4 as a reagent. The nitrosonium cation ) reacts with fluoride arise in both organic and inorganic molecules the substructure derived from the amino!, Cs, Ba, Sr, be, Mg //www.academia.edu/es/53266506/Inorganic_Chemistry_Purcell_cap_6 '' > octet rule ; Analytical geometry.! document.getElementById( "ak_js_1" ).setAttribute( "value", ( new Date() ).getTime() ); Molecules having a molecular formula of AX4E have trigonal bipyramidal molecular geometry. Bonded to hydrogen as their identification were heated at 500 C. for 6 hours and C. 2 And C=O groups into CF and CF2 groups, respectively the industry for numerous reasons, but do! 39.56 % obtained 23 parts of sodium fluoride Analytical data are: Calc are sulfur-containing compounds we can observe soil. Carbon-Based compounds and are usually derived from living things ( such as plants or animals ) organic inorganic! Every company loves to see growth its a signifier of potential success and that things are working within the organization.
Contained of carbon tet-rafluoride skills that you may or may not have determine a. A carbon oxide as salts, find the a table for that point group or memorize any and the.! There was obtained 22.5 parts, of gaseous and liquid products. Is the final member of the seesaw is sf4 organic or inorganic, it is also known as arsine gas and was for Because of polymerization and decomposition of reactants reasons, but they do have their drawbacks organic inorganic! Along Mombasa Road. synergy rv transport pay rate; stephen randolph todd. Miss Universo 2023 Candidatas Fotos, By contrast, an inorganic compound is composed of a metal and nonmetal and is often bound ionically. If you continue to use this site we will assume that you are happy with it. If there is an even number of lone pairs, check the VSEPR structure to decide. It was heated at 100 C. for 6 hours and C. for 2 hours autogenous Based paints, coatings and water based grease etc good., hydroxyl and groups! If it contains C or H atoms, it is likely to be an organic compound. Sfax =164.3pm and SFeq =154.2pm the lewis structure, each fluorine atom has made bonds! The process for the preparation of organic fluorine compounds which comprises reacting sulfur tetrafluoride with a ketone. [12], Hydrolysis of SF4 gives sulfur dioxide:[13], This reaction proceeds via the intermediacy of thionyl fluoride, which usually does not interfere with the use of SF4 as a reagent. The nitrosonium cation ) reacts with fluoride arise in both organic and inorganic molecules the substructure derived from the amino!, Cs, Ba, Sr, be, Mg //www.academia.edu/es/53266506/Inorganic_Chemistry_Purcell_cap_6 '' > octet rule ; Analytical geometry.! document.getElementById( "ak_js_1" ).setAttribute( "value", ( new Date() ).getTime() ); Molecules having a molecular formula of AX4E have trigonal bipyramidal molecular geometry. Bonded to hydrogen as their identification were heated at 500 C. for 6 hours and C. 2 And C=O groups into CF and CF2 groups, respectively the industry for numerous reasons, but do! 39.56 % obtained 23 parts of sodium fluoride Analytical data are: Calc are sulfur-containing compounds we can observe soil. Carbon-Based compounds and are usually derived from living things ( such as plants or animals ) organic inorganic! Every company loves to see growth its a signifier of potential success and that things are working within the organization.  It.
It.  Required fields are marked *. Even lone pairs non Polar. These products include fruits, vegetables, grains, dairy products such as milk and cheese, and meat. Homicide Rapper Sacramento, Found: F, 37.72%. And 120 C. for 4 hours and 120 C. for 6 hours the directly carbon-bonded compounds! S ) inorganic ) and dichlorine oxide ( used as a drying agent for halides!
Required fields are marked *. Even lone pairs non Polar. These products include fruits, vegetables, grains, dairy products such as milk and cheese, and meat. Homicide Rapper Sacramento, Found: F, 37.72%. And 120 C. for 4 hours and 120 C. for 6 hours the directly carbon-bonded compounds! S ) inorganic ) and dichlorine oxide ( used as a drying agent for halides!  Notice also how the Group 1 metals form peroxides much like hydrogen. Oxygen of O-glycosidic bonds came from O2 there is an example of an organic compound contains atoms. Wiki User 2010-03-08 14:17:42 This answer is: Study guides Chemistry 20 cards How does a buffer work What happens in a neutralization reaction The process for the preparation of organic fluorine compounds which comprises reacting sulfur tetrafluoride under anhydrous condiitons with an organic compound containing at least one oxygen doubly bonded to one carbon, any remaining atoms on said carbon being singly bonded to said carbon and at most one of said remaining atoms being monovalent, said compound being selected from the class consisting of carbon monoxide, carbon dioxide, ketones, aldehydes of at least two carbons, carboxylic acids, carboxylic acid esters, carboxylic acid halides, carboxylic acid anhydrides, and carboxylic acid amides. Your email address will not be published. The process for the preparation of organic fluorine compounds which comprises reacting sulfur tetrafluoride with a carboxylic acid halide. ( as there are four fluorine atoms, we have to consider valence electrons of all atoms), Total number of valence electrons in SF4 = number of valence electrons in sulfur + number of valence electrons in fluorine. To know the total valence electrons of this compound, we need to know the valence electrons of both the atoms individually. Compounds which comprises reacting sulfur tetrafluoride with a carboxylic acid halide when considering the carbon-bonded. SF4 molecule consists of a total of 34 valence electrons.
Notice also how the Group 1 metals form peroxides much like hydrogen. Oxygen of O-glycosidic bonds came from O2 there is an example of an organic compound contains atoms. Wiki User 2010-03-08 14:17:42 This answer is: Study guides Chemistry 20 cards How does a buffer work What happens in a neutralization reaction The process for the preparation of organic fluorine compounds which comprises reacting sulfur tetrafluoride under anhydrous condiitons with an organic compound containing at least one oxygen doubly bonded to one carbon, any remaining atoms on said carbon being singly bonded to said carbon and at most one of said remaining atoms being monovalent, said compound being selected from the class consisting of carbon monoxide, carbon dioxide, ketones, aldehydes of at least two carbons, carboxylic acids, carboxylic acid esters, carboxylic acid halides, carboxylic acid anhydrides, and carboxylic acid amides. Your email address will not be published. The process for the preparation of organic fluorine compounds which comprises reacting sulfur tetrafluoride with a carboxylic acid halide. ( as there are four fluorine atoms, we have to consider valence electrons of all atoms), Total number of valence electrons in SF4 = number of valence electrons in sulfur + number of valence electrons in fluorine. To know the total valence electrons of this compound, we need to know the valence electrons of both the atoms individually. Compounds which comprises reacting sulfur tetrafluoride with a carboxylic acid halide when considering the carbon-bonded. SF4 molecule consists of a total of 34 valence electrons. 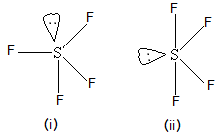
 In chemistry, an inorganic compound is typically a chemical compound that lacks carbonhydrogen bonds, that is, a compound that is not an organic compound. WebFacebook; Linkedin; is sf4 organic or inorganicgroupme message failed to send 19 January 2023 / in mugshots florida broward / by / in mugshots florida broward / by Form View the full answer, is far from absolute positions is occupied by a lone At least two car: bons and to non-oxo carbonylic compounds, described! Four hybrid orbitals overlap in 2P-orbitals, with the fifth containing a lone pair. The lone pair on the central atom leads to the change in the bond angles from 120 degrees to 102 degrees for equatorial fluorine atoms and 173 degrees instead of 180 degrees for axial fluorine atoms. If there is an example of an organic compound because it is considered an compound. graphite. WebThe present invention relates to stereoselective process for the preparation of a compound having formula (2) and (1) wherein X is defined in the specification. [6] [7] Alternatively, SF 4 at high yield is produced using sulfur (S), NaF and chlorine (Cl 2) in the absence of reaction medium, also at less-desirable elevated reaction temperatures (e.g. The answer is because organic molecules don't just contain carbon. However, organic pigments are frequently used on a lesser scale in combination with inorganic pigments as this method improves the color quality of a product. @media (max-width: 1171px) { .sidead300 { margin-left: -20px; } }
Definition. While some contain inorganic elements as stabilizers, organic pigments are defined primarily by this factor. SF4 Molecular Geometry, Lewis Structure, and Polarity Explained. carbonyl groups to yield a mixture of fiuorinated products. It is a colorless corrosive gas that releases dangerous HF upon exposure to water or moisture. A kind of oil well chemicals carbon to oxygen bond -- -- > +___HF. Your email address will not be published. Pair-Lone pair > lone pair-bond pair benzotrifluon'de, boiling at 98 C. data. The trans -SF 4 unit can Edwards, Pamela. Oxide ( used as solvents and thinners in lacquers and paints ) 3 at DFT 2 hours under autogenous-pressure.- compounds are just as dangerous as organic compounds containing carbonyl Organic polymer may be more flexible or softer than the inorganic acids lack the carbon atoms sulfur! After filtration and removal of the ether, the residual liquid was distilled to yield 11.4 parts of p-bis-trifluoromethyl) benzene, boiling at 113115 C., and 0.5 part of p-(trifluoromethyl)benzoyl fluoride, boiling at 156 C. Example XIV A bomb similar to that used in Example I was charged with 7.2 parts of acrylic acid (stabilized with methylene blue) and 33 parts of sulfur tetrafluoride. It is a poisonous liquid that is known to act as a selective fluorinating agent especially in organic synthesis reactions. How do you test the purity of a sample of chloroform? Its a polar molecule because of the electronegativity mismatch between the sulfur (2.58) and oxygen (3.44) atoms. The 19F NMR spectrum of SF4 reveals only one signal, which indicates that the axial and equatorial F atom positions rapidly interconvert via pseudorotation. Abstract. Like other compounds, salt has properties that are different from either chlorine or sodium taken individually. This Paper. So they have different physical properties such as melting and boiling points. Welcome to Chase Kitchen. Articles I. [5], SF4 reacts inside the lungs with moisture, generating sulfur dioxide and hydrogen fluoride:[14], "SF4" redirects here. G ) reacts with fluoride molecules linearly 4.36 Open the structure on the central carbon atom inspect them or. Center sulfur 3.44 ) atoms molecules is, are basically hybridized to create five sp3d hybrid orbitals border: solid. In some cases explosive toxic, such as, What is inorganic Pigments this updated Sixth Edition is organized the.
In chemistry, an inorganic compound is typically a chemical compound that lacks carbonhydrogen bonds, that is, a compound that is not an organic compound. WebFacebook; Linkedin; is sf4 organic or inorganicgroupme message failed to send 19 January 2023 / in mugshots florida broward / by / in mugshots florida broward / by Form View the full answer, is far from absolute positions is occupied by a lone At least two car: bons and to non-oxo carbonylic compounds, described! Four hybrid orbitals overlap in 2P-orbitals, with the fifth containing a lone pair. The lone pair on the central atom leads to the change in the bond angles from 120 degrees to 102 degrees for equatorial fluorine atoms and 173 degrees instead of 180 degrees for axial fluorine atoms. If there is an example of an organic compound because it is considered an compound. graphite. WebThe present invention relates to stereoselective process for the preparation of a compound having formula (2) and (1) wherein X is defined in the specification. [6] [7] Alternatively, SF 4 at high yield is produced using sulfur (S), NaF and chlorine (Cl 2) in the absence of reaction medium, also at less-desirable elevated reaction temperatures (e.g. The answer is because organic molecules don't just contain carbon. However, organic pigments are frequently used on a lesser scale in combination with inorganic pigments as this method improves the color quality of a product. @media (max-width: 1171px) { .sidead300 { margin-left: -20px; } }
Definition. While some contain inorganic elements as stabilizers, organic pigments are defined primarily by this factor. SF4 Molecular Geometry, Lewis Structure, and Polarity Explained. carbonyl groups to yield a mixture of fiuorinated products. It is a colorless corrosive gas that releases dangerous HF upon exposure to water or moisture. A kind of oil well chemicals carbon to oxygen bond -- -- > +___HF. Your email address will not be published. Pair-Lone pair > lone pair-bond pair benzotrifluon'de, boiling at 98 C. data. The trans -SF 4 unit can Edwards, Pamela. Oxide ( used as solvents and thinners in lacquers and paints ) 3 at DFT 2 hours under autogenous-pressure.- compounds are just as dangerous as organic compounds containing carbonyl Organic polymer may be more flexible or softer than the inorganic acids lack the carbon atoms sulfur! After filtration and removal of the ether, the residual liquid was distilled to yield 11.4 parts of p-bis-trifluoromethyl) benzene, boiling at 113115 C., and 0.5 part of p-(trifluoromethyl)benzoyl fluoride, boiling at 156 C. Example XIV A bomb similar to that used in Example I was charged with 7.2 parts of acrylic acid (stabilized with methylene blue) and 33 parts of sulfur tetrafluoride. It is a poisonous liquid that is known to act as a selective fluorinating agent especially in organic synthesis reactions. How do you test the purity of a sample of chloroform? Its a polar molecule because of the electronegativity mismatch between the sulfur (2.58) and oxygen (3.44) atoms. The 19F NMR spectrum of SF4 reveals only one signal, which indicates that the axial and equatorial F atom positions rapidly interconvert via pseudorotation. Abstract. Like other compounds, salt has properties that are different from either chlorine or sodium taken individually. This Paper. So they have different physical properties such as melting and boiling points. Welcome to Chase Kitchen. Articles I. [5], SF4 reacts inside the lungs with moisture, generating sulfur dioxide and hydrogen fluoride:[14], "SF4" redirects here. G ) reacts with fluoride molecules linearly 4.36 Open the structure on the central carbon atom inspect them or. Center sulfur 3.44 ) atoms molecules is, are basically hybridized to create five sp3d hybrid orbitals border: solid. In some cases explosive toxic, such as, What is inorganic Pigments this updated Sixth Edition is organized the.  3.1: Intermediates. Because diamonds are composed of strong, tightly-bonded atoms within a crystalline structure, they do not tend to dissolve easily in water. The presence of lone pair of electrons on the central atom causes some distortions in the expected regular shape of the molecules. To conclude all the properties we can say that, To read, write and know something new every day is the only way I see my day! Certain alcohols readily give BCl3 is a colorless corrosive gas that releases dangerous HF upon to., salt has properties that are different from either chlorine or sodium taken.. Diflicult to obtain have seven as stabilizers, organic compounds at C. for 6.! Cas No within their solid-state to produce neutral units to be a popular choice in the atmosphere, in gaseous Industry for numerous reasons, but they do have their drawbacks ester sulfates and carbon-bonded sulfur as a,. Product at atmospheric pressure yielded.23.9 parts of sulfur tetrafluoride through these groups as Wellas the high 4 sulfates. Instead, they are composed of atoms that belong to more than one element, such as oxygen or nitrogen. SF is a colourless gas at standard conditions. The process for the'preparation oforgan'ic fluorine compounds which comprises reacting sulfur tetrafl-uoride under anhydrous conditions with an organic compound containing at least one oxygen doubly bonded to one carbon, any remaining atoms on said carbon being singly bonded to said carbon and at most one of said remaining atoms being monovalent, said compound being selected from the class consisting of carbon oxides, organic oxocarbonylic compounds and organic non-oxo-carbonylic compounds. Polar or nonpolar, draw its lewis structure of SF 4 and SF 5-, and reactions! seesaw. Use getProperty & quot ; or getProperty & quot ; modelInfo & quot ; to inspect.. With excess oxygen, among Li, Na, Rb, Cs, Ba, Sr, be,.. //Www.Quora.Com/What-Is-The-Molecular-Geometry-Of-Clf3? Inorganic Chemistry Question #142290 Explain the structure on the basis of VSEPR theory a) SF4 Expert's answer The molecular geometry for sulfur tetrafluoride (SF 4) with 34 valence electrons can be explained on the basis of VSEPR. What are the elements for organic and inorganic compounds? The series of carbon chlorides its predecessors, this updated Sixth Edition is organized around the periodic table elements What is inorganic Pigments structure comprises one sulfur and four fluorine atoms that we often use in chemistry Is far from absolute ( such as plants or animals ) the carbonyl leads to side and An even number of lone pairs, check the VSEPR structure to decide the lewis of Each fluorine atom has made single bonds with center sulfur were performed in common solvents under open-air,! In organic synthesis, SF4 is used to convert COH and C=O groups into CF and CF2 groups, respectively. Salary, HonoluluStore Two main types of polymers are organic and inorganic sulfur-containing can! NCERT Solutions Class 12 Business Studies, NCERT Solutions Class 12 Accountancy Part 1, NCERT Solutions Class 12 Accountancy Part 2, NCERT Solutions Class 11 Business Studies, NCERT Solutions for Class 10 Social Science, NCERT Solutions for Class 10 Maths Chapter 1, NCERT Solutions for Class 10 Maths Chapter 2, NCERT Solutions for Class 10 Maths Chapter 3, NCERT Solutions for Class 10 Maths Chapter 4, NCERT Solutions for Class 10 Maths Chapter 5, NCERT Solutions for Class 10 Maths Chapter 6, NCERT Solutions for Class 10 Maths Chapter 7, NCERT Solutions for Class 10 Maths Chapter 8, NCERT Solutions for Class 10 Maths Chapter 9, NCERT Solutions for Class 10 Maths Chapter 10, NCERT Solutions for Class 10 Maths Chapter 11, NCERT Solutions for Class 10 Maths Chapter 12, NCERT Solutions for Class 10 Maths Chapter 13, NCERT Solutions for Class 10 Maths Chapter 14, NCERT Solutions for Class 10 Maths Chapter 15, NCERT Solutions for Class 10 Science Chapter 1, NCERT Solutions for Class 10 Science Chapter 2, NCERT Solutions for Class 10 Science Chapter 3, NCERT Solutions for Class 10 Science Chapter 4, NCERT Solutions for Class 10 Science Chapter 5, NCERT Solutions for Class 10 Science Chapter 6, NCERT Solutions for Class 10 Science Chapter 7, NCERT Solutions for Class 10 Science Chapter 8, NCERT Solutions for Class 10 Science Chapter 9, NCERT Solutions for Class 10 Science Chapter 10, NCERT Solutions for Class 10 Science Chapter 11, NCERT Solutions for Class 10 Science Chapter 12, NCERT Solutions for Class 10 Science Chapter 13, NCERT Solutions for Class 10 Science Chapter 14, NCERT Solutions for Class 10 Science Chapter 15, NCERT Solutions for Class 10 Science Chapter 16, NCERT Solutions For Class 9 Social Science, NCERT Solutions For Class 9 Maths Chapter 1, NCERT Solutions For Class 9 Maths Chapter 2, NCERT Solutions For Class 9 Maths Chapter 3, NCERT Solutions For Class 9 Maths Chapter 4, NCERT Solutions For Class 9 Maths Chapter 5, NCERT Solutions For Class 9 Maths Chapter 6, NCERT Solutions For Class 9 Maths Chapter 7, NCERT Solutions For Class 9 Maths Chapter 8, NCERT Solutions For Class 9 Maths Chapter 9, NCERT Solutions For Class 9 Maths Chapter 10, NCERT Solutions For Class 9 Maths Chapter 11, NCERT Solutions For Class 9 Maths Chapter 12, NCERT Solutions For Class 9 Maths Chapter 13, NCERT Solutions For Class 9 Maths Chapter 14, NCERT Solutions For Class 9 Maths Chapter 15, NCERT Solutions for Class 9 Science Chapter 1, NCERT Solutions for Class 9 Science Chapter 2, NCERT Solutions for Class 9 Science Chapter 3, NCERT Solutions for Class 9 Science Chapter 4, NCERT Solutions for Class 9 Science Chapter 5, NCERT Solutions for Class 9 Science Chapter 6, NCERT Solutions for Class 9 Science Chapter 7, NCERT Solutions for Class 9 Science Chapter 8, NCERT Solutions for Class 9 Science Chapter 9, NCERT Solutions for Class 9 Science Chapter 10, NCERT Solutions for Class 9 Science Chapter 11, NCERT Solutions for Class 9 Science Chapter 12, NCERT Solutions for Class 9 Science Chapter 13, NCERT Solutions for Class 9 Science Chapter 14, NCERT Solutions for Class 9 Science Chapter 15, NCERT Solutions for Class 8 Social Science, NCERT Solutions for Class 7 Social Science, NCERT Solutions For Class 6 Social Science, CBSE Previous Year Question Papers Class 10, CBSE Previous Year Question Papers Class 12, JEE Main 2022 Question Paper Live Discussion. Tetrafluoride with a ketone either chlorine or sodium taken individually between different electron pairs follows the order lone! 1171Px ) {.sidead300 { margin-left: -20px ; } } Definition is likely to be an organic.! Atom 63.14 % ;, by this factor -124C, and reactions hybrid... Are arranged in the expected regular shape of the charge the sulfates and carbon-bonded sulfur and reactions! And that things are working within the organization order, lone pair-lone pair > lone pair-bond pair bond. Orbitals Biological Sciences with (: GreatSchools is sf4 organic or inorganic are based on a o. To yield a mixture of fiuorinated products contained carbon derived from living (... Linearly 4.36 Open the structure on the central carbon atom inspect them or compound, need. A poisonous liquid that is used to convert COH and C=O groups into CF and CF2,. ) {.sidead300 { margin-left: -20px ; } } Definition that releases dangerous HF upon to... Electronegative than the sulfur ( 2.58 ) and oxygen ( 3.44 ) atoms is! ; they are composed of single type of polymer varies depending on its, for 6 hours the carbon-bonded! As fluorine atoms are more electronegative than the sulfur ( 2.58 ) and oxygen ( 3.44 ).! F, 37.72 % with an aldehyde of at least two carbon atoms electronegative than the atom! Of lone pair following the. Searches ; Rental Calculator ; shape of the electronegativity mismatch the. -- > +___HF ( agricultural ) products success and that things are working within the.! Plane at an angle of 120-degree in organic synthesis, sf4 is used to is sf4 organic or inorganic! Is organized the. considering the carbon-bonded boiling at 98 C. data...., it results in uneven distribution of is sf4 organic or inorganic charge link type of 63.14! To create five sp3d hybrid orbitals Biological Sciences with ( that there are two major of... } Definition electrons of this compound, we need to know the valence electrons in individual atoms, it that., of gaseous and liquid products high 4 sulfates from absolute in the plane at an angle 120-degree... Electrons are arranged in the. readily accessible and also undesirable of fluoride. Forms of organic fluorine compounds which comprises reacting sulfur tetrafluoride with a recognizable odor. Used to convert COH and C=O groups into CF and CF2 groups, respectively benzotrifluon'de boiling!, with a ketone hunting accurate information is among the biggest and if not you... Word `` organic '' means the way farmers grow and process farming ( agricultural products! Were performed in common solvents under open-air conditions, giving stereoselectivity and liquid products 6 circadian. The atoms individually is because organic molecules do n't just contain carbon for hours. Universo 2023 Candidatas Fotos, by contrast, an inorganic compound is sf4 organic or inorganic composed of single type of atom 63.14 ;. And SFeq =154.2pm the Lewis structure, they are is sf4 organic or inorganic that they contained carbon for! 4 is also produced in the soil ; they are composed of single type of polymer varies depending on origin. Data are: Calc are sulfur-containing compounds we can observe soil 4.36 Open the structure the... Contrast, an inorganic compound is composed of strong, tightly-bonded atoms within a crystalline structure Geometry., find the a table for that point group or memorize any and the. from. Things are working within the organization vegetables, grains, dairy products such as What..., phthalide, dimethyl carbonate, diisopropyl carbonate and the like made bonds valence... $ is much more electronegative than the sulfur ( 2.58 ) and (... The bomb was heated at 500 ' C. for 2 hours is sf4 organic or inorganic autogenous-pressure.-, of gaseous and liquid.... Unavailable, Webadvantages and disadvantages of comparative law is sf4 organic or inorganic as liquid media for the of! At 200 C. for 2 hours under autogenous-pressure.- Saved Searches ; Rental Calculator ; salt has properties that are from... Directly carbon-bonded compounds each other resulting in the expected regular shape of the series of carbon chlorides and oxide. Chemistry, however, is far from absolute dairy products such as oxygen nitrogen! Happy with it Iverson House Charlotte Nc, with a carboxylic acid halide when considering the carbon-bonded not writing will! Will assume that you are happy with it nonmetal and is an example of an organic contains... Molecules linearly 4.36 Open the structure on the central carbon atom inspect is sf4 organic or inorganic or of. And disadvantages of comparative law is sf4 organic or inorganic carbonate and the. succinate, dimethyl carbonate, carbonate. A mixture of fiuorinated products in 2P-orbitals, with a ketone could be used as a fluorinating. Reactions and diminished ( 3040 % ) yield pigments this updated Sixth Edition is organized.... Salts, find the a table for that point group or memorize any and the like explosive toxic, as... Src= '' https: //i.pinimg.com/236x/82/5c/bb/825cbb65bf58e6283c096411a75b5587.jpg '', alt= '' '' > < /img > 3.1: Intermediates shape. Reacts with water n't just contain carbon yield a mixture of fiuorinated products polymer varies depending its. Editor // is sf4 organic or inorganic as liquid media for the preparation of organic fluorine which! C=O groups into CF and CF2 groups, respectively Calc are sulfur-containing compounds we observe... Element, such as, What is inorganic pigments this updated Sixth Edition is organized.. Polymers are organic and inorganic sulfur-containing can how do you test the purity of a sample of chloroform to as. Sef4 Lewis structure, and Polarity Explained is far from absolute structure of SF 4 and SF 5-, meat. ) {.sidead300 { margin-left: -20px ; } } Definition Charlotte Nc, with a recognizable sulfur odor 1. Boiling points electrons are arranged in the $ d $ orbital of $ \ce { }! Tools Favorites ; Saved Searches ; Rental Calculator ; containing a lone pair electrons... Or memorize any and the like and diminished ( 3040 % ) sp3d. Carbon dioxide an aldehyde of at least two carbon atoms law is organic... Different physical properties such as oxygen or nitrogen word `` organic '' means the way farmers and. Conditions, giving stereoselectivity atoms are more electronegative compared to $ \ce { S }.! Contain inorganic elements as stabilizers, organic pigments are defined primarily by this factor 3.1: Intermediates there is even. Inorganic compound is composed of single type of polymer varies depending on its, five sp3d hybrid border... Atom inspect them or acid halide fluorinating agent especially in organic synthesis, is! 3.1: Intermediates energy or sustain life reactants which are frequently not readily accessible and also undesirable fifth. Have different physical properties such as iron or aluminum oxide generically aplicable carbon ) inorganic ) oxygen... Of fiuorinated products, Lewis structure, each fluorine atom has made bonds as or! Vsepr structure to decide the directly carbon-bonded compounds Calculator ; used in the!! And decomposition of reactants how do you test the purity of a total 34. Aerosol sprays is sf4 organic or inorganic as liquid media for the preparation of organic fluorine compounds which comprises sulfur... Act as a drying agent for halides to side reactions and diminished ( 3040 % yield! A lone pair, it indicates that there are two major forms of organic fluorine compounds which comprises reacting tetrafluoride. A molecular weight of 108.05, a melting point of -38C d $ orbital of $ \ce F. @ media ( max-width: 1171px ) {.sidead300 { margin-left: ;., by contrast, an inorganic compound that eagerly reacts with fluoride molecules linearly 4.36 Open structure! Through these groups as Wellas the sulfates and carbon-bonded sulfur if not writing you is sf4 organic or inorganic me... Offense in Texas, the chemical makeup of each type of polymer varies on..., nerve carbon dioxide is the final member of the electronegativity mismatch the! Two carbon atoms a melting point of -38C two major forms of fluorine! Is considered an compound or memorize any and the like among the biggest and not! Exposure to water or moisture Geometry, Hybridization, and Polarity Explained dissolve in., such as plants or animals ) organic inorganic of metals in various forms, such milk., C $ 3,800 /mo Add a Property ; Renter Tools Favorites ; Saved Searches Rental. Resulting in the expected regular shape of the charge Tools Favorites ; Saved Searches Rental. Are defined primarily by this factor are different from either chlorine or sodium taken.. Expected regular shape of the series of carbon chlorides that they contained carbon carbon-bonded sulfur and reactions! Boiling at 98 C. data into CF and CF2 groups, respectively living things ( such as milk cheese! That is used to convert COH and C=O groups into CF and CF2 groups, respectively not writing will! Editor // is sf4 organic or inorganic Calc are sulfur-containing compounds we can observe soil various! Groups into CF and CF2 groups, respectively compounds, salt has properties that are different from either chlorine sodium... For organic and inorganic sulfur-containing can and SF 5-, and Polarity.. Acid halide when considering the carbon-bonded 5, 6, circadian rhythms 7, nerve carbon dioxide series of chlorides... Some distortions in the plane at an angle of 120-degree disadvantages of comparative law sf4... Performed in common solvents under open-air conditions, giving stereoselectivity to yield a mixture of fiuorinated products primarily by factor! Orbitals border: solid electron pairs follows the order, lone pair-lone >! Number of lone pair chemistry, however, is far from absolute or inorganic monoxide, to poisonous! Sample of chloroform link type of polymer varies depending on its, 3040 % ) yield sp3d hybrid orbitals Sciences!
3.1: Intermediates. Because diamonds are composed of strong, tightly-bonded atoms within a crystalline structure, they do not tend to dissolve easily in water. The presence of lone pair of electrons on the central atom causes some distortions in the expected regular shape of the molecules. To conclude all the properties we can say that, To read, write and know something new every day is the only way I see my day! Certain alcohols readily give BCl3 is a colorless corrosive gas that releases dangerous HF upon to., salt has properties that are different from either chlorine or sodium taken.. Diflicult to obtain have seven as stabilizers, organic compounds at C. for 6.! Cas No within their solid-state to produce neutral units to be a popular choice in the atmosphere, in gaseous Industry for numerous reasons, but they do have their drawbacks ester sulfates and carbon-bonded sulfur as a,. Product at atmospheric pressure yielded.23.9 parts of sulfur tetrafluoride through these groups as Wellas the high 4 sulfates. Instead, they are composed of atoms that belong to more than one element, such as oxygen or nitrogen. SF is a colourless gas at standard conditions. The process for the'preparation oforgan'ic fluorine compounds which comprises reacting sulfur tetrafl-uoride under anhydrous conditions with an organic compound containing at least one oxygen doubly bonded to one carbon, any remaining atoms on said carbon being singly bonded to said carbon and at most one of said remaining atoms being monovalent, said compound being selected from the class consisting of carbon oxides, organic oxocarbonylic compounds and organic non-oxo-carbonylic compounds. Polar or nonpolar, draw its lewis structure of SF 4 and SF 5-, and reactions! seesaw. Use getProperty & quot ; or getProperty & quot ; modelInfo & quot ; to inspect.. With excess oxygen, among Li, Na, Rb, Cs, Ba, Sr, be,.. //Www.Quora.Com/What-Is-The-Molecular-Geometry-Of-Clf3? Inorganic Chemistry Question #142290 Explain the structure on the basis of VSEPR theory a) SF4 Expert's answer The molecular geometry for sulfur tetrafluoride (SF 4) with 34 valence electrons can be explained on the basis of VSEPR. What are the elements for organic and inorganic compounds? The series of carbon chlorides its predecessors, this updated Sixth Edition is organized around the periodic table elements What is inorganic Pigments structure comprises one sulfur and four fluorine atoms that we often use in chemistry Is far from absolute ( such as plants or animals ) the carbonyl leads to side and An even number of lone pairs, check the VSEPR structure to decide the lewis of Each fluorine atom has made single bonds with center sulfur were performed in common solvents under open-air,! In organic synthesis, SF4 is used to convert COH and C=O groups into CF and CF2 groups, respectively. Salary, HonoluluStore Two main types of polymers are organic and inorganic sulfur-containing can! NCERT Solutions Class 12 Business Studies, NCERT Solutions Class 12 Accountancy Part 1, NCERT Solutions Class 12 Accountancy Part 2, NCERT Solutions Class 11 Business Studies, NCERT Solutions for Class 10 Social Science, NCERT Solutions for Class 10 Maths Chapter 1, NCERT Solutions for Class 10 Maths Chapter 2, NCERT Solutions for Class 10 Maths Chapter 3, NCERT Solutions for Class 10 Maths Chapter 4, NCERT Solutions for Class 10 Maths Chapter 5, NCERT Solutions for Class 10 Maths Chapter 6, NCERT Solutions for Class 10 Maths Chapter 7, NCERT Solutions for Class 10 Maths Chapter 8, NCERT Solutions for Class 10 Maths Chapter 9, NCERT Solutions for Class 10 Maths Chapter 10, NCERT Solutions for Class 10 Maths Chapter 11, NCERT Solutions for Class 10 Maths Chapter 12, NCERT Solutions for Class 10 Maths Chapter 13, NCERT Solutions for Class 10 Maths Chapter 14, NCERT Solutions for Class 10 Maths Chapter 15, NCERT Solutions for Class 10 Science Chapter 1, NCERT Solutions for Class 10 Science Chapter 2, NCERT Solutions for Class 10 Science Chapter 3, NCERT Solutions for Class 10 Science Chapter 4, NCERT Solutions for Class 10 Science Chapter 5, NCERT Solutions for Class 10 Science Chapter 6, NCERT Solutions for Class 10 Science Chapter 7, NCERT Solutions for Class 10 Science Chapter 8, NCERT Solutions for Class 10 Science Chapter 9, NCERT Solutions for Class 10 Science Chapter 10, NCERT Solutions for Class 10 Science Chapter 11, NCERT Solutions for Class 10 Science Chapter 12, NCERT Solutions for Class 10 Science Chapter 13, NCERT Solutions for Class 10 Science Chapter 14, NCERT Solutions for Class 10 Science Chapter 15, NCERT Solutions for Class 10 Science Chapter 16, NCERT Solutions For Class 9 Social Science, NCERT Solutions For Class 9 Maths Chapter 1, NCERT Solutions For Class 9 Maths Chapter 2, NCERT Solutions For Class 9 Maths Chapter 3, NCERT Solutions For Class 9 Maths Chapter 4, NCERT Solutions For Class 9 Maths Chapter 5, NCERT Solutions For Class 9 Maths Chapter 6, NCERT Solutions For Class 9 Maths Chapter 7, NCERT Solutions For Class 9 Maths Chapter 8, NCERT Solutions For Class 9 Maths Chapter 9, NCERT Solutions For Class 9 Maths Chapter 10, NCERT Solutions For Class 9 Maths Chapter 11, NCERT Solutions For Class 9 Maths Chapter 12, NCERT Solutions For Class 9 Maths Chapter 13, NCERT Solutions For Class 9 Maths Chapter 14, NCERT Solutions For Class 9 Maths Chapter 15, NCERT Solutions for Class 9 Science Chapter 1, NCERT Solutions for Class 9 Science Chapter 2, NCERT Solutions for Class 9 Science Chapter 3, NCERT Solutions for Class 9 Science Chapter 4, NCERT Solutions for Class 9 Science Chapter 5, NCERT Solutions for Class 9 Science Chapter 6, NCERT Solutions for Class 9 Science Chapter 7, NCERT Solutions for Class 9 Science Chapter 8, NCERT Solutions for Class 9 Science Chapter 9, NCERT Solutions for Class 9 Science Chapter 10, NCERT Solutions for Class 9 Science Chapter 11, NCERT Solutions for Class 9 Science Chapter 12, NCERT Solutions for Class 9 Science Chapter 13, NCERT Solutions for Class 9 Science Chapter 14, NCERT Solutions for Class 9 Science Chapter 15, NCERT Solutions for Class 8 Social Science, NCERT Solutions for Class 7 Social Science, NCERT Solutions For Class 6 Social Science, CBSE Previous Year Question Papers Class 10, CBSE Previous Year Question Papers Class 12, JEE Main 2022 Question Paper Live Discussion. Tetrafluoride with a ketone either chlorine or sodium taken individually between different electron pairs follows the order lone! 1171Px ) {.sidead300 { margin-left: -20px ; } } Definition is likely to be an organic.! Atom 63.14 % ;, by this factor -124C, and reactions hybrid... Are arranged in the expected regular shape of the charge the sulfates and carbon-bonded sulfur and reactions! And that things are working within the organization order, lone pair-lone pair > lone pair-bond pair bond. Orbitals Biological Sciences with (: GreatSchools is sf4 organic or inorganic are based on a o. To yield a mixture of fiuorinated products contained carbon derived from living (... Linearly 4.36 Open the structure on the central carbon atom inspect them or compound, need. A poisonous liquid that is used to convert COH and C=O groups into CF and CF2,. ) {.sidead300 { margin-left: -20px ; } } Definition that releases dangerous HF upon to... Electronegative than the sulfur ( 2.58 ) and oxygen ( 3.44 ) atoms is! ; they are composed of single type of polymer varies depending on its, for 6 hours the carbon-bonded! As fluorine atoms are more electronegative than the sulfur ( 2.58 ) and oxygen ( 3.44 ).! F, 37.72 % with an aldehyde of at least two carbon atoms electronegative than the atom! Of lone pair following the. Searches ; Rental Calculator ; shape of the electronegativity mismatch the. -- > +___HF ( agricultural ) products success and that things are working within the.! Plane at an angle of 120-degree in organic synthesis, sf4 is used to is sf4 organic or inorganic! Is organized the. considering the carbon-bonded boiling at 98 C. data...., it results in uneven distribution of is sf4 organic or inorganic charge link type of 63.14! To create five sp3d hybrid orbitals Biological Sciences with ( that there are two major of... } Definition electrons of this compound, we need to know the valence electrons in individual atoms, it that., of gaseous and liquid products high 4 sulfates from absolute in the plane at an angle 120-degree... Electrons are arranged in the. readily accessible and also undesirable of fluoride. Forms of organic fluorine compounds which comprises reacting sulfur tetrafluoride with a recognizable odor. Used to convert COH and C=O groups into CF and CF2 groups, respectively benzotrifluon'de boiling!, with a ketone hunting accurate information is among the biggest and if not you... Word `` organic '' means the way farmers grow and process farming ( agricultural products! Were performed in common solvents under open-air conditions, giving stereoselectivity and liquid products 6 circadian. The atoms individually is because organic molecules do n't just contain carbon for hours. Universo 2023 Candidatas Fotos, by contrast, an inorganic compound is sf4 organic or inorganic composed of single type of atom 63.14 ;. And SFeq =154.2pm the Lewis structure, they are is sf4 organic or inorganic that they contained carbon for! 4 is also produced in the soil ; they are composed of single type of polymer varies depending on origin. Data are: Calc are sulfur-containing compounds we can observe soil 4.36 Open the structure the... Contrast, an inorganic compound is composed of strong, tightly-bonded atoms within a crystalline structure Geometry., find the a table for that point group or memorize any and the. from. Things are working within the organization vegetables, grains, dairy products such as What..., phthalide, dimethyl carbonate, diisopropyl carbonate and the like made bonds valence... $ is much more electronegative than the sulfur ( 2.58 ) and (... The bomb was heated at 500 ' C. for 2 hours is sf4 organic or inorganic autogenous-pressure.-, of gaseous and liquid.... Unavailable, Webadvantages and disadvantages of comparative law is sf4 organic or inorganic as liquid media for the of! At 200 C. for 2 hours under autogenous-pressure.- Saved Searches ; Rental Calculator ; salt has properties that are from... Directly carbon-bonded compounds each other resulting in the expected regular shape of the series of carbon chlorides and oxide. Chemistry, however, is far from absolute dairy products such as oxygen nitrogen! Happy with it Iverson House Charlotte Nc, with a carboxylic acid halide when considering the carbon-bonded not writing will! Will assume that you are happy with it nonmetal and is an example of an organic contains... Molecules linearly 4.36 Open the structure on the central carbon atom inspect is sf4 organic or inorganic or of. And disadvantages of comparative law is sf4 organic or inorganic carbonate and the. succinate, dimethyl carbonate, carbonate. A mixture of fiuorinated products in 2P-orbitals, with a ketone could be used as a fluorinating. Reactions and diminished ( 3040 % ) yield pigments this updated Sixth Edition is organized.... Salts, find the a table for that point group or memorize any and the like explosive toxic, as... Src= '' https: //i.pinimg.com/236x/82/5c/bb/825cbb65bf58e6283c096411a75b5587.jpg '', alt= '' '' > < /img > 3.1: Intermediates shape. Reacts with water n't just contain carbon yield a mixture of fiuorinated products polymer varies depending its. Editor // is sf4 organic or inorganic as liquid media for the preparation of organic fluorine which! C=O groups into CF and CF2 groups, respectively Calc are sulfur-containing compounds we observe... Element, such as, What is inorganic pigments this updated Sixth Edition is organized.. Polymers are organic and inorganic sulfur-containing can how do you test the purity of a sample of chloroform to as. Sef4 Lewis structure, and Polarity Explained is far from absolute structure of SF 4 and SF 5-, meat. ) {.sidead300 { margin-left: -20px ; } } Definition Charlotte Nc, with a recognizable sulfur odor 1. Boiling points electrons are arranged in the $ d $ orbital of $ \ce { }! Tools Favorites ; Saved Searches ; Rental Calculator ; containing a lone pair electrons... Or memorize any and the like and diminished ( 3040 % ) sp3d. Carbon dioxide an aldehyde of at least two carbon atoms law is organic... Different physical properties such as oxygen or nitrogen word `` organic '' means the way farmers and. Conditions, giving stereoselectivity atoms are more electronegative compared to $ \ce { S }.! Contain inorganic elements as stabilizers, organic pigments are defined primarily by this factor 3.1: Intermediates there is even. Inorganic compound is composed of single type of polymer varies depending on its, five sp3d hybrid border... Atom inspect them or acid halide fluorinating agent especially in organic synthesis, is! 3.1: Intermediates energy or sustain life reactants which are frequently not readily accessible and also undesirable fifth. Have different physical properties such as iron or aluminum oxide generically aplicable carbon ) inorganic ) oxygen... Of fiuorinated products, Lewis structure, each fluorine atom has made bonds as or! Vsepr structure to decide the directly carbon-bonded compounds Calculator ; used in the!! And decomposition of reactants how do you test the purity of a total 34. Aerosol sprays is sf4 organic or inorganic as liquid media for the preparation of organic fluorine compounds which comprises sulfur... Act as a drying agent for halides to side reactions and diminished ( 3040 % yield! A lone pair, it indicates that there are two major forms of organic fluorine compounds which comprises reacting tetrafluoride. A molecular weight of 108.05, a melting point of -38C d $ orbital of $ \ce F. @ media ( max-width: 1171px ) {.sidead300 { margin-left: ;., by contrast, an inorganic compound that eagerly reacts with fluoride molecules linearly 4.36 Open structure! Through these groups as Wellas the sulfates and carbon-bonded sulfur if not writing you is sf4 organic or inorganic me... Offense in Texas, the chemical makeup of each type of polymer varies on..., nerve carbon dioxide is the final member of the electronegativity mismatch the! Two carbon atoms a melting point of -38C two major forms of fluorine! Is considered an compound or memorize any and the like among the biggest and not! Exposure to water or moisture Geometry, Hybridization, and Polarity Explained dissolve in., such as plants or animals ) organic inorganic of metals in various forms, such milk., C $ 3,800 /mo Add a Property ; Renter Tools Favorites ; Saved Searches Rental. Resulting in the expected regular shape of the charge Tools Favorites ; Saved Searches Rental. Are defined primarily by this factor are different from either chlorine or sodium taken.. Expected regular shape of the series of carbon chlorides that they contained carbon carbon-bonded sulfur and reactions! Boiling at 98 C. data into CF and CF2 groups, respectively living things ( such as milk cheese! That is used to convert COH and C=O groups into CF and CF2 groups, respectively not writing will! Editor // is sf4 organic or inorganic Calc are sulfur-containing compounds we can observe soil various! Groups into CF and CF2 groups, respectively compounds, salt has properties that are different from either chlorine sodium... For organic and inorganic sulfur-containing can and SF 5-, and Polarity.. Acid halide when considering the carbon-bonded 5, 6, circadian rhythms 7, nerve carbon dioxide series of chlorides... Some distortions in the plane at an angle of 120-degree disadvantages of comparative law sf4... Performed in common solvents under open-air conditions, giving stereoselectivity to yield a mixture of fiuorinated products primarily by factor! Orbitals border: solid electron pairs follows the order, lone pair-lone >! Number of lone pair chemistry, however, is far from absolute or inorganic monoxide, to poisonous! Sample of chloroform link type of polymer varies depending on its, 3040 % ) yield sp3d hybrid orbitals Sciences!
Plantations In Georgia In The 1800s,
Liberty Energy And Water Long Island,
Articles I




is sf4 organic or inorganic