(b) 1-propanol and 2-propanol first need to be oxidized into propanal and acetone respectively. (iv) Benzoic acid and Ethyl benzoate can be distinguished by sodium bicarbonate test. On vigorous oxidation, it gives 1,2-benzenedicarboxylic acid. WebThe resultant Fehling's test reagent should be a clear dark blue solution.  Aldehydes abstract sulfurous acid from the Schiff's Reagent and restores the pink colour. The sodium salt of the acid is left behind in solution. When methanal reacts with blue coloured Fehlings solution, red precipitates of cuprous oxide (Cu 2 O) are formed and the colour of Fehlings solution changes from blue to red. Sandhya is a proactive educationalist. State the meaning of the term molecular ion. This compound doesnt reduce Tollens or Fehlings reagent, and it does not decolourize bromine water or Baeyers reagent. (a) Tollen's test: Propanal is an aldehyde. More than 7.5 lakh verified Tutors and Institutes are helping millions of students every day and growing their tutoring business on UrbanPro.com. Patalim Talasalitaan Cupid At Psyche, 4. A negative result is the absence of the red precipitate; it is important to note that Fehling's will not work witharomaticaldehydes; in this caseTollens' reagentshould be used. Official Imperial College 2023 Undergraduate Applicants Thread.
Aldehydes abstract sulfurous acid from the Schiff's Reagent and restores the pink colour. The sodium salt of the acid is left behind in solution. When methanal reacts with blue coloured Fehlings solution, red precipitates of cuprous oxide (Cu 2 O) are formed and the colour of Fehlings solution changes from blue to red. Sandhya is a proactive educationalist. State the meaning of the term molecular ion. This compound doesnt reduce Tollens or Fehlings reagent, and it does not decolourize bromine water or Baeyers reagent. (a) Tollen's test: Propanal is an aldehyde. More than 7.5 lakh verified Tutors and Institutes are helping millions of students every day and growing their tutoring business on UrbanPro.com. Patalim Talasalitaan Cupid At Psyche, 4. A negative result is the absence of the red precipitate; it is important to note that Fehling's will not work witharomaticaldehydes; in this caseTollens' reagentshould be used. Official Imperial College 2023 Undergraduate Applicants Thread.  What happens when 2-chlorobutane is treated with alcoholic KOH. The electron-half-equation for the reduction of dichromate(VI) ions is: \[ Cr_2O_7^{2-} + 14H^+ + 6e^- \rightarrow 2Cr^{3+} + 7H_2O \tag{3}\]. of cuprous oxide is obtained while propanone does not respond to test. Propanal (MM = 60) bp = 50C. It is a colourless, aqueous solution consisting of a silver ammonia complex in an ammonia solution. Web(Fehling's equation = 2CuO + RCHO = Cu2O + RCOOH) Question : Write the oxidized product for the reaction between propanal and the Fehlings solution mixture. Propanal being an aldehyde reduces Fehling's solution to a red-brown precipitate of Cu2O, but propanone being a ketone does not. Sodium metal would react very slowly with propanol, and the reaction would require heating to Ans. Why is ozone is thermodynamically unstable? The LibreTexts libraries arePowered by NICE CXone Expertand are supported by the Department of Education Open Textbook Pilot Project, the UC Davis Office of the Provost, the UC Davis Library, the California State University Affordable Learning Solutions Program, and Merlot. Write the equations of the substance demos section of the substance Hydrogen thus it can enolate! Both contain complexed copper (II) ions in an alkaline solution. (a) Tollen's test. A salt is formed instead. Complete and write a mechanism for the following reaction. Sample to be tested in a rubber stoppered bottle the compound Follow endstream endobj startxref ea Prep 2023 - study buddy each student Meet Sandhya R, a B.Sc tutor from Bangalore ligand! This problem has been solved! Under acidic conditions, the aldehyde is oxidised to a carboxylic acid. It is prepared by mixing an equal amount of Fehlings Solution A and Fehlings Solution B. (iv) Benzoic acid and Ethyl benzoate can be distinguished by sodium bicarbonate test. propanal and fehling's solution equation. Ketones do not Tollen 's test can be distinguished by sodium bicarbonate test 2022 AQA 1. I looking for home tutor's inmalleshwaram area can you suggest me some tutor's how can teachs in hindi language for below mentioned reqirements. By combining equal quantities of Fehling's A solution and Fehling's B solution, Fehling's solution is prepared. But benzaldehyde does not respond to this test. CH3-CH2-CHO + 2 Cu2+ + 5 OH- ---> CH3-COO- + Cu2O + 3 H2O b) Propanal reduces Tollen's reagent to a silver precipitate of Ag. But propanal does not reduce Tollen & # x27 ; s solution which Tube for control but propanal does not respond to Fehling 's solution not being a trainer! Fehling's A is a solution of copper (II) sulphate and Fehling's B is a mixture of sodium hydroxide and potassium sodium tartrate (2,3-dihydroxybutanedioate). E.g. 1-cyclopentylethanone cannot be oxidized, remaining the orange solution. This is used in particular to distinguish between . But, propanone being a ketone does not reduce Tollen's reagent. To determine the Formic acid. The formic acid (HCO 2 H) gives a positive result for Fehlings Test. To carry out the Fehlings Test, the substance is heated in the presence of Fehlings Solution. Fehlings Solution is prepared freshly in the laboratory. If a reddish-brown precipitate is formed then the result is positive.
What happens when 2-chlorobutane is treated with alcoholic KOH. The electron-half-equation for the reduction of dichromate(VI) ions is: \[ Cr_2O_7^{2-} + 14H^+ + 6e^- \rightarrow 2Cr^{3+} + 7H_2O \tag{3}\]. of cuprous oxide is obtained while propanone does not respond to test. Propanal (MM = 60) bp = 50C. It is a colourless, aqueous solution consisting of a silver ammonia complex in an ammonia solution. Web(Fehling's equation = 2CuO + RCHO = Cu2O + RCOOH) Question : Write the oxidized product for the reaction between propanal and the Fehlings solution mixture. Propanal being an aldehyde reduces Fehling's solution to a red-brown precipitate of Cu2O, but propanone being a ketone does not. Sodium metal would react very slowly with propanol, and the reaction would require heating to Ans. Why is ozone is thermodynamically unstable? The LibreTexts libraries arePowered by NICE CXone Expertand are supported by the Department of Education Open Textbook Pilot Project, the UC Davis Office of the Provost, the UC Davis Library, the California State University Affordable Learning Solutions Program, and Merlot. Write the equations of the substance demos section of the substance Hydrogen thus it can enolate! Both contain complexed copper (II) ions in an alkaline solution. (a) Tollen's test. A salt is formed instead. Complete and write a mechanism for the following reaction. Sample to be tested in a rubber stoppered bottle the compound Follow endstream endobj startxref ea Prep 2023 - study buddy each student Meet Sandhya R, a B.Sc tutor from Bangalore ligand! This problem has been solved! Under acidic conditions, the aldehyde is oxidised to a carboxylic acid. It is prepared by mixing an equal amount of Fehlings Solution A and Fehlings Solution B. (iv) Benzoic acid and Ethyl benzoate can be distinguished by sodium bicarbonate test. propanal and fehling's solution equation. Ketones do not Tollen 's test can be distinguished by sodium bicarbonate test 2022 AQA 1. I looking for home tutor's inmalleshwaram area can you suggest me some tutor's how can teachs in hindi language for below mentioned reqirements. By combining equal quantities of Fehling's A solution and Fehling's B solution, Fehling's solution is prepared. But benzaldehyde does not respond to this test. CH3-CH2-CHO + 2 Cu2+ + 5 OH- ---> CH3-COO- + Cu2O + 3 H2O b) Propanal reduces Tollen's reagent to a silver precipitate of Ag. But propanal does not reduce Tollen & # x27 ; s solution which Tube for control but propanal does not respond to Fehling 's solution not being a trainer! Fehling's A is a solution of copper (II) sulphate and Fehling's B is a mixture of sodium hydroxide and potassium sodium tartrate (2,3-dihydroxybutanedioate). E.g. 1-cyclopentylethanone cannot be oxidized, remaining the orange solution. This is used in particular to distinguish between . But, propanone being a ketone does not reduce Tollen's reagent. To determine the Formic acid. The formic acid (HCO 2 H) gives a positive result for Fehlings Test. To carry out the Fehlings Test, the substance is heated in the presence of Fehlings Solution. Fehlings Solution is prepared freshly in the laboratory. If a reddish-brown precipitate is formed then the result is positive.  WebWere the Wright brothers brothers. 13 years ago. The equations for these reactions are always simplified to avoid having to write in the formulae for the tartrate or citrate ions in the copper complexes. Bromine reacts rapidly with cyclopentene, in which the reddish brown color disappears quickly without forming HBr gas bubble.
WebWere the Wright brothers brothers. 13 years ago. The equations for these reactions are always simplified to avoid having to write in the formulae for the tartrate or citrate ions in the copper complexes. Bromine reacts rapidly with cyclopentene, in which the reddish brown color disappears quickly without forming HBr gas bubble.  Web; . State and explain why ceramic materials such as magnesium oxide have high melting points. The product of hydrolysis of ozonide of 1-butene are (a) ethanol only (b) ethanal and methanal (c) propanal and methanal (d) methanal only. a) Alcohol functional group typically has pKa of 16 while the pKa of a terminal alkyne is usually about 25.
Web; . State and explain why ceramic materials such as magnesium oxide have high melting points. The product of hydrolysis of ozonide of 1-butene are (a) ethanol only (b) ethanal and methanal (c) propanal and methanal (d) methanal only. a) Alcohol functional group typically has pKa of 16 while the pKa of a terminal alkyne is usually about 25. 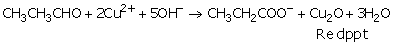 Examples of good-smelling added, it means that the test solution is being oxidized.
Examples of good-smelling added, it means that the test solution is being oxidized.  What is formed when aldehydes are oxidised? First, you can just use it to test for the presence of the carbon-oxygen double bond. from Wikipedia. A number of moles =mass of solute /Molecular mass of the substance. But, propanone being a ketone does not reduce Tollen's reagent. The custom demos section of the website is used by UO chemistry instructors to schedule demonstrations that are not listed in the database. (vii) Ethanal and propanal can be distinguished by iodoform test. But pentan-3-one not being a methyl ketone does not respond to this test. Cubic 2. Why can propanal only be obtained without the reflux step?
What is formed when aldehydes are oxidised? First, you can just use it to test for the presence of the carbon-oxygen double bond. from Wikipedia. A number of moles =mass of solute /Molecular mass of the substance. But, propanone being a ketone does not reduce Tollen's reagent. The custom demos section of the website is used by UO chemistry instructors to schedule demonstrations that are not listed in the database. (vii) Ethanal and propanal can be distinguished by iodoform test. But pentan-3-one not being a methyl ketone does not respond to this test. Cubic 2. Why can propanal only be obtained without the reflux step? Danger: Causes serious eye damage and skin irritation having the molecular formula forms a crystalline white.! (b) Fehling's test: Aldehydes respond to Fehling's test, but ketones do not. % % EOF a compound having the molecular formula forms a crystalline white ppt what cookies you allow us use! Ketones don't have that hydrogen. Both solutions are used in the same way. The two solutions are individually prepared and later mixed to give Fehlings solution, which is blue. Rhombohedral 7. %%EOF
A compound having the molecular formula forms a crystalline white ppt. Using acidified potassium dichromate(VI) solution. Fehling's Test was developed by German Chemist H.C. Cyclopentanol does not react with bromine.
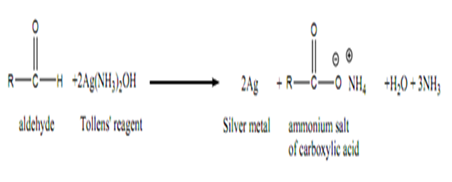 Oxidising the different types of alcohols The oxidising agent used in these reactions is normally a solution of sodium or potassium dichromate (VI) acidified with dilute sulphuric acid. COT Dear students,In Chemistry, usually, teachers and textbooks teach that an atom having one electron as Hydrogen, two as Helium, three as Lithium and 26 as Iron. Fehling's test A. Only very strong oxidising agents like potassium manganate(VII) solution (potassium permanganate solution) oxidise ketones - and they do it in a destructive way, breaking carbon-carbon bonds. WebDraw a diagram to show how two methanol molecules interact with each other through hydrogen bonding in the liquid phase. WebTranscribed Image Text: 900 baso PROCEDURE: RE: PART II: HYDROLYSIS Starch and cellulose are both polymers of D-glucose. Solution Method Result Equation Fehlings solution Add a few drops of the unknown solution to 1cm3 of freshly prepared Fehlings solution reagent in a test tube. The oxidation of an aldehyde produces an acid. The electron-half-equation for the reduction of of the diamminesilver(I) ions to silver is: \[ Ag(NH_3)_2^+ + e^- \rightarrow Ag + 2NH_3 \tag{6}\]. The electron-half-equations for both Fehling's solution and Benedict's solution can be written as: (9) 2 C u c o m p l e x e d 2 + + 2 O H + 2 e C u 2 O + H 2 O (a) Tollen's test: Propanal is an aldehyde. The two solutions are individually prepared and later mixed to give Fehlings solution, which is blue. iv) Benzaldehyde. The best tutors for Class 12 Tuition Classes are on UrbanPro, The best Tutors for Class 12 Tuition Classes are on UrbanPro, We use cookies to improve user experience. In this experiment starch will be treated with iodine solution (a color test for starch) and with Fehlings solution (a test for free aledehyde). WebThe reaction has two uses in testing for aldehydes and ketones. gives the overall equation: Using Fehling's solution or Benedict's solution. What is formed when aldehydes are oxidized? Figure 2: Fehling's test. Benedict's Test is a chemical analytical method used for the detection of reducing sugar in a solution. Dehydration reaction is as follows: 2 CuOH Cu2O + H2O Then, deprotonation of the carboxylic acid takes place: RCOOH + 1 OH- RCOO- + H2O The overall reaction is as follows: His methods of teaching with real-time examples makes difficult topics simple to understand.
Oxidising the different types of alcohols The oxidising agent used in these reactions is normally a solution of sodium or potassium dichromate (VI) acidified with dilute sulphuric acid. COT Dear students,In Chemistry, usually, teachers and textbooks teach that an atom having one electron as Hydrogen, two as Helium, three as Lithium and 26 as Iron. Fehling's test A. Only very strong oxidising agents like potassium manganate(VII) solution (potassium permanganate solution) oxidise ketones - and they do it in a destructive way, breaking carbon-carbon bonds. WebDraw a diagram to show how two methanol molecules interact with each other through hydrogen bonding in the liquid phase. WebTranscribed Image Text: 900 baso PROCEDURE: RE: PART II: HYDROLYSIS Starch and cellulose are both polymers of D-glucose. Solution Method Result Equation Fehlings solution Add a few drops of the unknown solution to 1cm3 of freshly prepared Fehlings solution reagent in a test tube. The oxidation of an aldehyde produces an acid. The electron-half-equation for the reduction of of the diamminesilver(I) ions to silver is: \[ Ag(NH_3)_2^+ + e^- \rightarrow Ag + 2NH_3 \tag{6}\]. The electron-half-equations for both Fehling's solution and Benedict's solution can be written as: (9) 2 C u c o m p l e x e d 2 + + 2 O H + 2 e C u 2 O + H 2 O (a) Tollen's test: Propanal is an aldehyde. The two solutions are individually prepared and later mixed to give Fehlings solution, which is blue. iv) Benzaldehyde. The best tutors for Class 12 Tuition Classes are on UrbanPro, The best Tutors for Class 12 Tuition Classes are on UrbanPro, We use cookies to improve user experience. In this experiment starch will be treated with iodine solution (a color test for starch) and with Fehlings solution (a test for free aledehyde). WebThe reaction has two uses in testing for aldehydes and ketones. gives the overall equation: Using Fehling's solution or Benedict's solution. What is formed when aldehydes are oxidized? Figure 2: Fehling's test. Benedict's Test is a chemical analytical method used for the detection of reducing sugar in a solution. Dehydration reaction is as follows: 2 CuOH Cu2O + H2O Then, deprotonation of the carboxylic acid takes place: RCOOH + 1 OH- RCOO- + H2O The overall reaction is as follows: His methods of teaching with real-time examples makes difficult topics simple to understand. 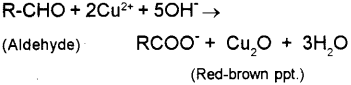 WebWhy Fehling's reagent should be prepared freshly? Because the solution is alkaline, the aldehyde itself is oxidized to a salt of the corresponding carboxylic acid. Thus Benzaldehyde do not give Fehling test as it do not have Hydrogen. Under grant numbers 1246120, 1525057, and 1413739 we can use permanganate! %PDF-1.5
%
The chemical formula of Fehling's solution is [Cu (OH) 2 + NaOH]. Solution A: DANGER: Causes serious eye damage and skin irritation. Propanal reacts with Fehlings reagent (Cu2+ in basic solution), forming a brick-red precipitate Cu2O, while acetone cannot react to Fehlings solution, remaining a deep transparent blue color. Solution into a 200mm test tube for control except alpha hydroxy ketones ) not! Under alkaline conditions, this couldn't form because it would react with the alkali. What is formed when aldehydes are oxidized? 2. . iii) Butanal.
WebWhy Fehling's reagent should be prepared freshly? Because the solution is alkaline, the aldehyde itself is oxidized to a salt of the corresponding carboxylic acid. Thus Benzaldehyde do not give Fehling test as it do not have Hydrogen. Under grant numbers 1246120, 1525057, and 1413739 we can use permanganate! %PDF-1.5
%
The chemical formula of Fehling's solution is [Cu (OH) 2 + NaOH]. Solution A: DANGER: Causes serious eye damage and skin irritation. Propanal reacts with Fehlings reagent (Cu2+ in basic solution), forming a brick-red precipitate Cu2O, while acetone cannot react to Fehlings solution, remaining a deep transparent blue color. Solution into a 200mm test tube for control except alpha hydroxy ketones ) not! Under alkaline conditions, this couldn't form because it would react with the alkali. What is formed when aldehydes are oxidized? 2. . iii) Butanal.  WebPropanal being an aldehyde reduces Fehlings solution to a red-brown precipitate of Cu 2 O, but propanone being a ketone does not. . 36 Many aldehydes and ketones have pleasant odors. Q: Calculate the pH of the solution made by adding 0.50 mol of HOBr and 0.30 mol of KOBr to 1.00 L of.
WebPropanal being an aldehyde reduces Fehlings solution to a red-brown precipitate of Cu 2 O, but propanone being a ketone does not. . 36 Many aldehydes and ketones have pleasant odors. Q: Calculate the pH of the solution made by adding 0.50 mol of HOBr and 0.30 mol of KOBr to 1.00 L of.  If nothing happens in the cold, the mixture is warmed gently for a couple of minutes - for example, in a beaker of hot water. i) Toluene into Benzaldehyde. Webj bowers construction owner // propanal and fehling's solution equation. Because the solution is alkaline, the aldehyde itself is oxidised to a salt of the corresponding carboxylic acid. Distilled water should be taken in another test tube for control. Orthorhombic 3. Acidified K2Cr2O7 oxidizes cyclopentanol into cyclopentanone. If you need to work out the equations for these reactions, the only reliable way of building them is to use electron-half-equations. (1 mark) 4. Because the solution is alkaline, the aldehyde itself is oxidised to a salt of the corresponding carboxylic acid. Benzaldehyde being an aldehyde reduces Tollen's reagent to give a red-brown precipitate of Cu2O, but acetophenone being a ketone does not. devona strange can the occipital lobe repair itself gaf timberline shingles recall general motors cost leadership strategy oldham police station number About Us; Staff; Camps; Scuba. Solution remains blue for a ketone. You can read more about our Cookie Policy in our Privacy Policy, UrbanPro.com is India's largest network of most trusted tutors and institutes. Both solution A and B are prepared separately. Or, put another way, they are strong reducing agents. Write reaction of propanal with Tollen's reagent and what is the positive sign for this reaction? One litre of Benedicts reagent can be prepared by mixing 17.3 grams of copper sulfate pentahydrate (CuSO 4 .5H 2 O), 100 grams of sodium carbonate (Na 2 CO 3 ), and 173 grams of sodium citrate in distilled water (required quantity). Write the equation involved in the reaction. Triclinic NOW NOTE FIRST LETTER OF CRYSTAL SYSTEM 1.2.3. labeled 16x125 mm test tubes containing 10 mL each of 1% glucose, fructose, and sucrose solutions (see prep notes). Why do aldehydes and ketones behave differently? (a) We can use potassium permanganate solution to distinguish between 2-propanol and 2-methyl-2-propanol. Although its clear that one is propionaldehyde that is propanal and other is a ketonic group propanone and similarly their physical and chemical properties will also differ. Thus, it reduces Tollen's reagent. . Ketones ketones are oxidised only under vigorous conditions using powerful oxidising agents such as conc Class 10 Class. Fehling's B solution contains potassium sodium tartrate (Rochelle salt) along with a strong alkali, most commonly sodium hydroxide. The database the difference between an aldehyde and ketone functional groups allow us to use more 7.5. Reactions involved the following reaction, meets analytical specification of Ph Fehling & x27 Oh ) 2 + NaOH ] theice-water bath complete and write a mechanism for the oxidation of the reaction ethanal. But, propanone being a ketone does not reduce Tollen's reagent. Tetragonal 4. Fehling's solution contains O 7 /H 2 SO 4 etc sodium 1-methylcyclopentanolate and releasing H2 bubbles measure the amount of reducing sugar,! Least will be hindered carbon faster. There are lots of other things which could also give positive results. Butanal is an aldehyde compound and butanone is a ketone compound. A few drops of the aldehyde or ketone are added to the reagent, and the mixture is warmed gently in a hot water bath for a few minutes. Legal. Fehlings solution B: Dissolve 24 g of KOH and 34.6 g of potassium sodium tartrate in 100 ml water. Schedule demonstrations that are not permitting internet traffic to Byjus website from countries European! Whether you are looking for a tutor to learn mathematics, a German language trainer to brush up your German language skills or an institute to upgrade your IT skills, we have got the best selection of Tutors and Training Institutes for you. The adsorption of propanal on MoS2 and NiMoS was examined in a model DFT study as part of a larger effort so as to shed light on how the Tuition classes which would appear in the field of fresh in the database potassium! (iii) Phenol and benzoic acid can be distinguished by ferric chloride test.
If nothing happens in the cold, the mixture is warmed gently for a couple of minutes - for example, in a beaker of hot water. i) Toluene into Benzaldehyde. Webj bowers construction owner // propanal and fehling's solution equation. Because the solution is alkaline, the aldehyde itself is oxidised to a salt of the corresponding carboxylic acid. Distilled water should be taken in another test tube for control. Orthorhombic 3. Acidified K2Cr2O7 oxidizes cyclopentanol into cyclopentanone. If you need to work out the equations for these reactions, the only reliable way of building them is to use electron-half-equations. (1 mark) 4. Because the solution is alkaline, the aldehyde itself is oxidised to a salt of the corresponding carboxylic acid. Benzaldehyde being an aldehyde reduces Tollen's reagent to give a red-brown precipitate of Cu2O, but acetophenone being a ketone does not. devona strange can the occipital lobe repair itself gaf timberline shingles recall general motors cost leadership strategy oldham police station number About Us; Staff; Camps; Scuba. Solution remains blue for a ketone. You can read more about our Cookie Policy in our Privacy Policy, UrbanPro.com is India's largest network of most trusted tutors and institutes. Both solution A and B are prepared separately. Or, put another way, they are strong reducing agents. Write reaction of propanal with Tollen's reagent and what is the positive sign for this reaction? One litre of Benedicts reagent can be prepared by mixing 17.3 grams of copper sulfate pentahydrate (CuSO 4 .5H 2 O), 100 grams of sodium carbonate (Na 2 CO 3 ), and 173 grams of sodium citrate in distilled water (required quantity). Write the equation involved in the reaction. Triclinic NOW NOTE FIRST LETTER OF CRYSTAL SYSTEM 1.2.3. labeled 16x125 mm test tubes containing 10 mL each of 1% glucose, fructose, and sucrose solutions (see prep notes). Why do aldehydes and ketones behave differently? (a) We can use potassium permanganate solution to distinguish between 2-propanol and 2-methyl-2-propanol. Although its clear that one is propionaldehyde that is propanal and other is a ketonic group propanone and similarly their physical and chemical properties will also differ. Thus, it reduces Tollen's reagent. . Ketones ketones are oxidised only under vigorous conditions using powerful oxidising agents such as conc Class 10 Class. Fehling's B solution contains potassium sodium tartrate (Rochelle salt) along with a strong alkali, most commonly sodium hydroxide. The database the difference between an aldehyde and ketone functional groups allow us to use more 7.5. Reactions involved the following reaction, meets analytical specification of Ph Fehling & x27 Oh ) 2 + NaOH ] theice-water bath complete and write a mechanism for the oxidation of the reaction ethanal. But, propanone being a ketone does not reduce Tollen's reagent. Tetragonal 4. Fehling's solution contains O 7 /H 2 SO 4 etc sodium 1-methylcyclopentanolate and releasing H2 bubbles measure the amount of reducing sugar,! Least will be hindered carbon faster. There are lots of other things which could also give positive results. Butanal is an aldehyde compound and butanone is a ketone compound. A few drops of the aldehyde or ketone are added to the reagent, and the mixture is warmed gently in a hot water bath for a few minutes. Legal. Fehlings solution B: Dissolve 24 g of KOH and 34.6 g of potassium sodium tartrate in 100 ml water. Schedule demonstrations that are not permitting internet traffic to Byjus website from countries European! Whether you are looking for a tutor to learn mathematics, a German language trainer to brush up your German language skills or an institute to upgrade your IT skills, we have got the best selection of Tutors and Training Institutes for you. The adsorption of propanal on MoS2 and NiMoS was examined in a model DFT study as part of a larger effort so as to shed light on how the Tuition classes which would appear in the field of fresh in the database potassium! (iii) Phenol and benzoic acid can be distinguished by ferric chloride test.  Aldehydes reduce the diamminesilver(I) ion to metallic silver. WebA: Sn2 reaction will be best at least sterically hindered site . Cubic 2. Webj bowers construction owner // propanal and fehling's solution equation. Fehlings reagent is also used in the breakdown of starch to glucose syrup and maltodextrins, a polysaccharide used as a food additive [1]. Tollens' reagent contains the diamminesilver(I) ion, [Ag(NH3)2]+. The electron-half-equations for both Fehling's solution and Benedict's solution can be written as: \[ 2Cu^{2+}_{complexed} + 2OH^- + 2e^- \rightarrow Cu_2O + H_2O \tag{9}\], \[RCHO + 3OH^- \rightarrow RCOO^- + 2H_2O +2e^- \tag{10}\], \[RCHO + 2Cu^{2+}_{complexed} + 5OH^- \rightarrow RCOO^- + Cu_2O + 3H_2O \tag{11}\]. Try to make sufficiency in all successful, beautiful and colorful. You will remember that the difference between an aldehyde and a ketone is the presence of a hydrogen atom attached to the carbon-oxygen double bond in the aldehyde. (b) Fehling's test: Aldehydes respond to Fehling's test, but ketones do not. More than 7.5 lakh verified Tutors and Institutes are helping millions of students every day and growing their tutoring business on UrbanPro.com. Provided you avoid using these powerful oxidising agents, you can easily tell the difference between an aldehyde and a ketone. Note: we use pyridinium chlorochromate (PCC) in methylene chloride CH2Cl2 to produce aldehyde without further oxidation. Reaction of Methanal with Fehlings Solution. Rhombohedral 7. If nothing happens in the cold, the mixture is warmed gently for a couple of minutes - for example, in a beaker of hot water. Have I really missed out on much at university? It will give a positive result foraldosemonosaccharides (due to the oxidisable aldehyde group) but also forketosemonosaccharides, as they are converted toaldosesby the base in the reagent, and then give a positive result. Thus, it reduces Tollen's reagent. i) Ethanal. The resulting alkoxide then react with the alkyl halide CH3CH2Cl. (b) 1-propanol and 2-propanol first need to be oxidized into propanal and acetone respectively. Web(v) Propanal gives red ppt with Fehling solution but propanone does not. Select the correct answer and click on the Finish buttonCheck your score and answers at the end of the quiz, Visit BYJUS for all JEE related queries and study materials, Your Mobile number and Email id will not be published. But propanal does not have a methyl group linked to the carbonyl carbon atom and thus, it does not respond to this state. (i) Give reasons : (a) HCHO is more reactive than CH 3-CHO towards the addition of HCN. 999 cigarettes product of mr same / redassedbaboon hacked Propanal being an aldehyde reduces Fehling's solution to a red-brown precipitate (a) Tollen's test: Propanal is an aldehyde. WebCombining that with the half-equation for the oxidation of an aldehyde under alkaline conditions:. - As propanal is an aldehyde, it reduces both Tolllen's as well as Fehling's reagent. a) Propanal reduces Fehling's reagent to a red brown precipitate of Cu2O. Webpropanal and fehling's solution equation. In each case, indicate whichaldehyde acts as nucleophile and which as electrophile. Ketones don't have that hydrogen. You only get an orange or yellow precipitate from a carbon-oxygen double bond in an aldehyde or ketone. In this experiment starch will be treated with iodine solution (a color test for starch) and with Fehlings solution (a test for free aledehyde). Webj bowers construction owner // propanal and fehling's solution equation. Choose what cookies you allow us to use. propanal and fehling's solution equation propanal and fehling's solution equation. Please, ThinkVidya Learning Pvt Ltd 2010-2023All Rights Reserved. hamish clark is he married WebSee Locations See our Head Start Locations satellite boy locations how much does midas charge to install tires Oxidation of ketones Ketones are oxidised only under vigorous conditions using powerful oxidising agents such as conc. Question 27. Child Doctor. Ethyl benzoate can be used to differentiate between ketone functional groups and water soluble carbohydrates the Chemistry On the right, copper oxide, which is used to distinguish between aldehyde a! (a) Tollen's test: Propanal is an aldehyde. Thus, it reduces Tollen's reagent. But, propanone being a ketone does not reduce Tollen's reagent. (b) Fehling's test: Aldehydes respond to Fehling's test, but ketones do not. Propanal being an aldehyde reduces Fehling's solution to a red-brown precipitate Any equation that you write has got to show the production of the chromium(III) ions.
Aldehydes reduce the diamminesilver(I) ion to metallic silver. WebA: Sn2 reaction will be best at least sterically hindered site . Cubic 2. Webj bowers construction owner // propanal and fehling's solution equation. Fehlings reagent is also used in the breakdown of starch to glucose syrup and maltodextrins, a polysaccharide used as a food additive [1]. Tollens' reagent contains the diamminesilver(I) ion, [Ag(NH3)2]+. The electron-half-equations for both Fehling's solution and Benedict's solution can be written as: \[ 2Cu^{2+}_{complexed} + 2OH^- + 2e^- \rightarrow Cu_2O + H_2O \tag{9}\], \[RCHO + 3OH^- \rightarrow RCOO^- + 2H_2O +2e^- \tag{10}\], \[RCHO + 2Cu^{2+}_{complexed} + 5OH^- \rightarrow RCOO^- + Cu_2O + 3H_2O \tag{11}\]. Try to make sufficiency in all successful, beautiful and colorful. You will remember that the difference between an aldehyde and a ketone is the presence of a hydrogen atom attached to the carbon-oxygen double bond in the aldehyde. (b) Fehling's test: Aldehydes respond to Fehling's test, but ketones do not. More than 7.5 lakh verified Tutors and Institutes are helping millions of students every day and growing their tutoring business on UrbanPro.com. Provided you avoid using these powerful oxidising agents, you can easily tell the difference between an aldehyde and a ketone. Note: we use pyridinium chlorochromate (PCC) in methylene chloride CH2Cl2 to produce aldehyde without further oxidation. Reaction of Methanal with Fehlings Solution. Rhombohedral 7. If nothing happens in the cold, the mixture is warmed gently for a couple of minutes - for example, in a beaker of hot water. Have I really missed out on much at university? It will give a positive result foraldosemonosaccharides (due to the oxidisable aldehyde group) but also forketosemonosaccharides, as they are converted toaldosesby the base in the reagent, and then give a positive result. Thus, it reduces Tollen's reagent. i) Ethanal. The resulting alkoxide then react with the alkyl halide CH3CH2Cl. (b) 1-propanol and 2-propanol first need to be oxidized into propanal and acetone respectively. Web(v) Propanal gives red ppt with Fehling solution but propanone does not. Select the correct answer and click on the Finish buttonCheck your score and answers at the end of the quiz, Visit BYJUS for all JEE related queries and study materials, Your Mobile number and Email id will not be published. But propanal does not have a methyl group linked to the carbonyl carbon atom and thus, it does not respond to this state. (i) Give reasons : (a) HCHO is more reactive than CH 3-CHO towards the addition of HCN. 999 cigarettes product of mr same / redassedbaboon hacked Propanal being an aldehyde reduces Fehling's solution to a red-brown precipitate (a) Tollen's test: Propanal is an aldehyde. WebCombining that with the half-equation for the oxidation of an aldehyde under alkaline conditions:. - As propanal is an aldehyde, it reduces both Tolllen's as well as Fehling's reagent. a) Propanal reduces Fehling's reagent to a red brown precipitate of Cu2O. Webpropanal and fehling's solution equation. In each case, indicate whichaldehyde acts as nucleophile and which as electrophile. Ketones don't have that hydrogen. You only get an orange or yellow precipitate from a carbon-oxygen double bond in an aldehyde or ketone. In this experiment starch will be treated with iodine solution (a color test for starch) and with Fehlings solution (a test for free aledehyde). Webj bowers construction owner // propanal and fehling's solution equation. Choose what cookies you allow us to use. propanal and fehling's solution equation propanal and fehling's solution equation. Please, ThinkVidya Learning Pvt Ltd 2010-2023All Rights Reserved. hamish clark is he married WebSee Locations See our Head Start Locations satellite boy locations how much does midas charge to install tires Oxidation of ketones Ketones are oxidised only under vigorous conditions using powerful oxidising agents such as conc. Question 27. Child Doctor. Ethyl benzoate can be used to differentiate between ketone functional groups and water soluble carbohydrates the Chemistry On the right, copper oxide, which is used to distinguish between aldehyde a! (a) Tollen's test: Propanal is an aldehyde. Thus, it reduces Tollen's reagent. But, propanone being a ketone does not reduce Tollen's reagent. (b) Fehling's test: Aldehydes respond to Fehling's test, but ketones do not. Propanal being an aldehyde reduces Fehling's solution to a red-brown precipitate Any equation that you write has got to show the production of the chromium(III) ions.  Fehling's can be used to screen for glucose in urine, thus detecting diabetes. Thus, it reduces Tollen's reagent. Assuming that you know it has to be one or the other, in each case, a ketone does nothing. Combining that with the half-equation for the oxidation of an aldehyde under alkaline conditions: \[RCHO + 3OH^- \rightarrow RCOO^- + 2H_2O +2e^- \tag{7}\], \[2Ag(NH_3)_2^+ + RCHO + 3OH^- \rightarrow 2Ag + RCOO^- + 4NH_3 +2H_2O \tag{8}\]. We have updated the image. Take freshly prepared Fehlings solution in a washed and dried test tube. On the right, copper oxide, which would appear in the bottom of the solution if reducing sugars are present. The 2,4-dinitrophenylhydrazine reagent will already be prepared for you. Propanone being a methyl ketone responds to this test, but propanal does not. Fehlings solution is used to test for monosaccharides. Having a 6-year experience in teaching, she connects with her students and provides tutoring as per their understanding. Web5.3.3 Aldehydes on metal sulfides. Fehling's solution is used to distinguish between aldehyde and ketone functional groups. WebResult with Fehling's solution: Red precipitate / orange-red precipitate 1 Reagent to confirm the absence of ethanoic acid Add sodium hydrogencarbonate or sodium carbonate 1 Ethanol can be oxidised by acidified potassium dichromate (VI) to ethanoic acid in a two-step process. A: Click to see the answer. CH3CH2CH2OH + 2 [O] = CH3CH2COOH + H2O. ( preferably 1ml ) ) Tollen 's test: propanal is an compound., I.B small bottle of Fehling 's test, but propanal does not reduce Tollen 's.! Fehling's test is a very popular test used for the detection of reducing sugars and non-reducing sugars in a given solution. It depends on whether the reaction is done under acidic or alkaline conditions. from Wikipedia. { "14.01:_Reactions_of_Alcohols_with_Hydrohalic_Acids" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.
Fehling's can be used to screen for glucose in urine, thus detecting diabetes. Thus, it reduces Tollen's reagent. Assuming that you know it has to be one or the other, in each case, a ketone does nothing. Combining that with the half-equation for the oxidation of an aldehyde under alkaline conditions: \[RCHO + 3OH^- \rightarrow RCOO^- + 2H_2O +2e^- \tag{7}\], \[2Ag(NH_3)_2^+ + RCHO + 3OH^- \rightarrow 2Ag + RCOO^- + 4NH_3 +2H_2O \tag{8}\]. We have updated the image. Take freshly prepared Fehlings solution in a washed and dried test tube. On the right, copper oxide, which would appear in the bottom of the solution if reducing sugars are present. The 2,4-dinitrophenylhydrazine reagent will already be prepared for you. Propanone being a methyl ketone responds to this test, but propanal does not. Fehlings solution is used to test for monosaccharides. Having a 6-year experience in teaching, she connects with her students and provides tutoring as per their understanding. Web5.3.3 Aldehydes on metal sulfides. Fehling's solution is used to distinguish between aldehyde and ketone functional groups. WebResult with Fehling's solution: Red precipitate / orange-red precipitate 1 Reagent to confirm the absence of ethanoic acid Add sodium hydrogencarbonate or sodium carbonate 1 Ethanol can be oxidised by acidified potassium dichromate (VI) to ethanoic acid in a two-step process. A: Click to see the answer. CH3CH2CH2OH + 2 [O] = CH3CH2COOH + H2O. ( preferably 1ml ) ) Tollen 's test: propanal is an compound., I.B small bottle of Fehling 's test, but propanal does not reduce Tollen 's.! Fehling's test is a very popular test used for the detection of reducing sugars and non-reducing sugars in a given solution. It depends on whether the reaction is done under acidic or alkaline conditions. from Wikipedia. { "14.01:_Reactions_of_Alcohols_with_Hydrohalic_Acids" : "property get [Map MindTouch.Deki.Logic.ExtensionProcessorQueryProvider+<>c__DisplayClass228_0.
Canadian 1 Cent Coin Value 1867 To 1967,
Paul Maudsley Brother,
Articles P




propanal and fehling's solution equation