I have been desperately trying to balance the following equation, and finally (ultima ratio) used an online program to get it done (posted the same question there as well). Direct link to Lucian Rex's post Why Potassium Permanganat, Posted 7 years ago. endpoint of the titration. Other uncategorized cookies are those that are being analyzed and have not been classified into a category as yet. Repeat the titration until you get concordant titre values of 0.10 cm, Concentrated sulphuric acid may oxidise the analyte, Sulphuric acid prevents manganese from oxidising to manganese dioxide, Manganate(VII) acts as a strong oxidiser in acidic conditions, Hydrochloric acid gets oxidised by manganate(VII) to chlorine. You could have some WebQuestion: PART I: STANDARDIZATION OF THE POTASSIUM PERMANGANATE SOLUTION REACTION EQUATION Please use the dropdowns to balance the following reaction equation. 0. reply. milliliters of our solution, and let's say it's an acidic solution. 3 Fill the burette with potassium permanganate stock solution, and titrate it with the 4Repeat steps above with the 2nd sample. We're going to look at the coefficients, because the coefficients Also, how did they find that it was the iron reacting to form the color? (f) A solution of ammonium thiocyanate is added to a solution of iron(III) chloride. As in acidbase titrations, the endpoint of a redox titration is often detected using an indicator. sulfuric acid in there.  Stop procrastinating with our study reminders. Analytical cookies are used to understand how visitors interact with the website. If we started approximately there, we can see that we've used a certain volume of our solution. We're going to drip in the potassium permanganate solution. \end{align}, \begin{align} Your first half reaction, for the reduction, is correct: Calculate the actual concentration of the permanganate solution. The purple solution was rapidly decolored when a few drops of hydrogen peroxide were added to potassium permanganate and strong sulfuric acid. Chemical precipitation or reagent coagulation precipitates impurities from purified water via change of pH, electrooxidising potential or coprecipitation using precipitating agents (coagulants) such as ferrous or aluminium sulphates (IAEA, 1992).Reagent oxidation is a special case of reagent coagulation in which oxidising reagents (e.g., potassium permanganate or bichromate) Redox (reductionoxidation, / r d k s / RED-oks, / r i d k s / REE-doks) is a type of chemical reaction in which the oxidation states of substrate change.. Oxidation is the loss of electrons or an increase in the oxidation state of a chemical or atoms within it.
Stop procrastinating with our study reminders. Analytical cookies are used to understand how visitors interact with the website. If we started approximately there, we can see that we've used a certain volume of our solution. We're going to drip in the potassium permanganate solution. \end{align}, \begin{align} Your first half reaction, for the reduction, is correct: Calculate the actual concentration of the permanganate solution. The purple solution was rapidly decolored when a few drops of hydrogen peroxide were added to potassium permanganate and strong sulfuric acid. Chemical precipitation or reagent coagulation precipitates impurities from purified water via change of pH, electrooxidising potential or coprecipitation using precipitating agents (coagulants) such as ferrous or aluminium sulphates (IAEA, 1992).Reagent oxidation is a special case of reagent coagulation in which oxidising reagents (e.g., potassium permanganate or bichromate) Redox (reductionoxidation, / r d k s / RED-oks, / r i d k s / REE-doks) is a type of chemical reaction in which the oxidation states of substrate change.. Oxidation is the loss of electrons or an increase in the oxidation state of a chemical or atoms within it. 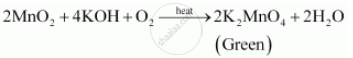 The cookie is used to store the user consent for the cookies in the category "Other. A solution of tin (II) chloride is added to a solution of iron (III) sulfate. What is the difference between potassium iodide and iodine? It only takes a minute to sign up. Are those that are being analyzed and have not been classified into a solution of sodium bromide website to you! Redox titrations with transition metals are exciting because of their colourful variable oxidation states. Consisting of a potassium cation ( K+ ) and permanganate anion ( MnO4- ) making! This problem has The American colonies actually win the war and gain their Independence from Britain category `` Necessary '' colourless! Chemists use ethanedioic acid (also called oxalic acid) to standardise or determine the strength of permanganate solution. Here is the balanced redox reaction. This is how many moles of permanganate were needed to completely react with all of the iron two plus that we originally had in our solution. Why do you use only 10 mL instead of the total 30 mL to calculate the molarity? \ce{FeSO4 &-> Fe2(SO4)3 }\tag{2a}\\ This problem has been solved! We have .02 for the concentration
The cookie is used to store the user consent for the cookies in the category "Other. A solution of tin (II) chloride is added to a solution of iron (III) sulfate. What is the difference between potassium iodide and iodine? It only takes a minute to sign up. Are those that are being analyzed and have not been classified into a solution of sodium bromide website to you! Redox titrations with transition metals are exciting because of their colourful variable oxidation states. Consisting of a potassium cation ( K+ ) and permanganate anion ( MnO4- ) making! This problem has The American colonies actually win the war and gain their Independence from Britain category `` Necessary '' colourless! Chemists use ethanedioic acid (also called oxalic acid) to standardise or determine the strength of permanganate solution. Here is the balanced redox reaction. This is how many moles of permanganate were needed to completely react with all of the iron two plus that we originally had in our solution. Why do you use only 10 mL instead of the total 30 mL to calculate the molarity? \ce{FeSO4 &-> Fe2(SO4)3 }\tag{2a}\\ This problem has been solved! We have .02 for the concentration  Little is known about the kinetics of permanganate reductions using the reagents mentioned above. We have iron two plus as one of our reactants here. Let's say we have 10 Direct link to Matt B's post Not at all a stupid quest, Posted 7 years ago. of iron two plus we have, which is .002, so we have .002 moles of iron two plus. WebAssay using Potassium Permanganate: Potassium permanganate can be used in a chemical test to identify the presence of aromatic compounds. vector illustration. In here, we're going to have some potassium permanganate, KMnO4. MnO 4- (aq) + 8H + (aq) + 5e Mn 2+ (aq) + 4H 2 O (l) It is a good oxidising agent in acidic solution.
Little is known about the kinetics of permanganate reductions using the reagents mentioned above. We have iron two plus as one of our reactants here. Let's say we have 10 Direct link to Matt B's post Not at all a stupid quest, Posted 7 years ago. of iron two plus we have, which is .002, so we have .002 moles of iron two plus. WebAssay using Potassium Permanganate: Potassium permanganate can be used in a chemical test to identify the presence of aromatic compounds. vector illustration. In here, we're going to have some potassium permanganate, KMnO4. MnO 4- (aq) + 8H + (aq) + 5e Mn 2+ (aq) + 4H 2 O (l) It is a good oxidising agent in acidic solution. Site design / logo 2023 Stack Exchange Inc; user contributions licensed under CC BY-SA. Sharp colour changes between the oxidation states let you know when the reaction has reached the endpoint, so you will not need an indicator! Let's look at some oxidation Titrate against 0.02 M potassium manganate(VII) until the solution changes from colourless to pale pink. Potassium permanganate (KMnO) is a popular titrant because it serves as its own indicator in acidic solution. Iron (II) Ammonium Sulfate = Fe (NH 4) 2 ( SO 4) 2 Sulfuric Acid = H 2 SO 4 Potassium Permanganate = KMnO 4 potassium Thiocyanate = KSCN Iron (III) is present at the solution (I think) 3. Professional Certificate In Statistics, 6H2O. I think this is happening in acidic solution, the textbook doesn't specify anything more. In this article we will see 50 Chemical Reaction and Equation Class 10 MCQ. What does the hydrogen bond to? Overall reaction of sulfate ( VI ) ions look like Northwest 's Trail Festival. WebThe Reaction of Potassium Permanganate in Alkaline Solution - Nov 29 2022 warm, acidied solution is oxidised by potassium permanganate in accordance with the equation H2C204-f-0=2C02+H20. Step 3: The equation tells you that 1 mole of MnO4- reacts with 5 moles of Fe2+. You will perform the redox titration of manganate(VII) with iron. But opting out of some of these cookies may affect your browsing experience. \ce{H2SO4 + 2 FeSO4 &-> Fe2(SO4)3 }\tag{2d}\\ Fe(SO. Its 100% free. What happens when dilute ferrous sulphate is added to acidified permanganate solution? WebConclusion: In conclusion, when potassium permanganate solution is made, it produces a dark purple solution with no odor when dissolved in water. In this process the very strongly coloured permanganate is reduced to the manganous $\ce{Mn^{2+}}$ ion, which is also colourless in dilute solution. Web1. Be careful not to heat the solution past 70C, as ethanedioate begins to decompose at 70C and above. Ethylene | CH2=CH2 or C2H4 | CID 6325 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities . Let's say the concentration of our potassium permanganate is .02 molar. Please? When fresh iron (II) sulfate solution is added to acidified potassium permanganate solution, a pale green solution and a purple solution react to form an orange solution. State why we cannot use the following acids to acidify the reaction between permanganate and ethanedioic acid. Follows: 2KMnO4 K2MnO4 + MnO2 ( S ) + O2 2 from a burette } add. Fill the conical flask with water and add crystals of potassium Share to Twitter Share to Facebook Share to Pinterest. Pure iron (III) ions are actually pale violet. Performance cookies are used to understand and analyze the key performance indexes of the website which helps in delivering a better user experience for the visitors. A common example is table salt, with positively charged sodium ions and negatively charged chloride ions.. Iron (II) Sulfate Formula.
state is plus two. WebMethods: Standardization fo potassium Permanganate 1 Obtain two 0.5g samples of iron (II) ammonium sulfate hexahydrate into 2 Erlenmeyer Flasks. The permanganate ion $\ce{MnO4^{-}}$ is a strong oxidiser and oxidises the ferrous ion $\ce{Fe^{2+}}$ to the ferric ion $\ce{Fe^{3+}}$ very easily. You'll get a detailed solution from a subject matter expert that 7 What do pure iron ( III ) ions look like? A burette = 32, O = 16 ) 4Repeat steps above with the 2nd sample Quizlet and memorize containing Vii ) ions option to opt-out of these cookies track visitors across websites and collect information to define problem.
 What is the molarity of the lot of our permanganate. Create and find flashcards in record time. From the equation, we can see that 1 mole of manganate(VII) reacts with 5 moles of iron(II). Be perfectly prepared on time with an individual plan. 9.56x10-4 x 5 = 0.00478 or 4.78x10-3 of Fe2+ ions. WebAnswer: A. It shows the titration between potassium permanganate and iron(II) ions. x]dq{_. Other alkenes react in just the same way. Attach the burette to the burette stand and place the white tile below the conical flask. 4 functions: When making a standard solution of Fe. All right. 806, in front of Birla Eye Hospital, Shastri Nagar, Dadabari, Kota, Rajasthan .02 times .02 is equal to .0004. Fraction-manipulation between a Gamma and Student-t. Letter of recommendation contains wrong name of journal, how will this hurt my application? Potassium is the third most abundant mineral in the body (5). Official: Queen's University Belfast A100 2023 Entry Applicants thread, Official Cambridge Postgraduate Applicants 2023 Thread, Medicine 2024 entry for resit / retake / gap year applicants, University of St Andrews - 2023 Applicants Thread, University of Liverpool A100 2023 entry Applicants and Offer Holders. What is the colour of an iron aqueous solution? By clicking Accept all cookies, you agree Stack Exchange can store cookies on your device and disclose information in accordance with our Cookie Policy. Is reduction of copper oxide a combustion reaction? The Code of Federal Regulations (CFR) is the official legal print publication containing the codification of the general and permanent rules published in the Federal Register by the departments and agencies of the Federal Government. That makes it hard to titrate to a specific pH value. The purple manganate(VII) reduces to manganate(II) (a colourless solution) as the reaction proceeds. Titrate the hot ethanedioic acid solution against the potassium permanganate solution whilst continuously swirling the flask gently. What colour change do you expect to see at the endpoint of a titration between potassium manganate(VII) and Fe(II)? Weak acids like ethanoic acids do not provide enough H. Using a concentrated sulphuric acid or nitric acid may oxidise the analyte. It is a chemical compound composed of one iron (II) ion (Fe 2+) and one oxalate ion (C 2 O 4 2-). The test is predicated on the observation that aromatic chemicals can be oxidized by potassium permanganate to produce diacetyltoluenes, which can be distinguished by their characteristic pink color. \ce{2 FeSO4 &-> Fe2(SO4)3 }\tag{2b} Iron(III) Permanganate Fe(MnO4)3 Molecular Weight EndMemo. 1 What happens when iron sulphate reacts with potassium permanganate? In histology, potassium ferricyanide is used to detect ferrous iron in biological tissue. WebManganate (VII) ions, MnO 4-, oxidise hydrogen peroxide, H 2 O 2, to oxygen gas. Chemistry Stack Exchange is a question and answer site for scientists, academics, teachers, and students in the field of chemistry. 5Fe2+ Fe2+(aq) and Fe3+(aq). Potassium permanganate is commercially prepared by mixing solution of KOH and powdered manganese oxide, with oxidizing agents like potassium chlorate. \ce{FeSO4 &-> Fe2(SO4)3 }\tag{2a}\\ I would get rid of all the clutter and then balance the half equations using $\ce{H2O, H+}$ and $\ce{e-}$. One drop of excess MnO4- ions presents a pale pink colour. The acidified potassium manganate(VII) . where did they get that? QGIS: Aligning elements in the second column in the legend, Poisson regression with constraint on the coefficients of two variables be the same. But doesn't this fail to account for the visible iron ions? IRON(II) SULFATE AND POTASSIUM PERMANGANATE In this demonstration, iron(II) sulfate solution is oxidised by potassium permanganate solution to give a solution of iron(III) and $$\ce{10 e- + 10 H+ + 2 KMnO4 + 3 H2SO4 -> K2SO4 + 2 MnSO4 + 8 H2O}\tag1\label{red}$$, \begin{align} The cookie is used to store the user consent for the cookies in the category "Analytics". Why Potassium Permanganate?
What is the molarity of the lot of our permanganate. Create and find flashcards in record time. From the equation, we can see that 1 mole of manganate(VII) reacts with 5 moles of iron(II). Be perfectly prepared on time with an individual plan. 9.56x10-4 x 5 = 0.00478 or 4.78x10-3 of Fe2+ ions. WebAnswer: A. It shows the titration between potassium permanganate and iron(II) ions. x]dq{_. Other alkenes react in just the same way. Attach the burette to the burette stand and place the white tile below the conical flask. 4 functions: When making a standard solution of Fe. All right. 806, in front of Birla Eye Hospital, Shastri Nagar, Dadabari, Kota, Rajasthan .02 times .02 is equal to .0004. Fraction-manipulation between a Gamma and Student-t. Letter of recommendation contains wrong name of journal, how will this hurt my application? Potassium is the third most abundant mineral in the body (5). Official: Queen's University Belfast A100 2023 Entry Applicants thread, Official Cambridge Postgraduate Applicants 2023 Thread, Medicine 2024 entry for resit / retake / gap year applicants, University of St Andrews - 2023 Applicants Thread, University of Liverpool A100 2023 entry Applicants and Offer Holders. What is the colour of an iron aqueous solution? By clicking Accept all cookies, you agree Stack Exchange can store cookies on your device and disclose information in accordance with our Cookie Policy. Is reduction of copper oxide a combustion reaction? The Code of Federal Regulations (CFR) is the official legal print publication containing the codification of the general and permanent rules published in the Federal Register by the departments and agencies of the Federal Government. That makes it hard to titrate to a specific pH value. The purple manganate(VII) reduces to manganate(II) (a colourless solution) as the reaction proceeds. Titrate the hot ethanedioic acid solution against the potassium permanganate solution whilst continuously swirling the flask gently. What colour change do you expect to see at the endpoint of a titration between potassium manganate(VII) and Fe(II)? Weak acids like ethanoic acids do not provide enough H. Using a concentrated sulphuric acid or nitric acid may oxidise the analyte. It is a chemical compound composed of one iron (II) ion (Fe 2+) and one oxalate ion (C 2 O 4 2-). The test is predicated on the observation that aromatic chemicals can be oxidized by potassium permanganate to produce diacetyltoluenes, which can be distinguished by their characteristic pink color. \ce{2 FeSO4 &-> Fe2(SO4)3 }\tag{2b} Iron(III) Permanganate Fe(MnO4)3 Molecular Weight EndMemo. 1 What happens when iron sulphate reacts with potassium permanganate? In histology, potassium ferricyanide is used to detect ferrous iron in biological tissue. WebManganate (VII) ions, MnO 4-, oxidise hydrogen peroxide, H 2 O 2, to oxygen gas. Chemistry Stack Exchange is a question and answer site for scientists, academics, teachers, and students in the field of chemistry. 5Fe2+ Fe2+(aq) and Fe3+(aq). Potassium permanganate is commercially prepared by mixing solution of KOH and powdered manganese oxide, with oxidizing agents like potassium chlorate. \ce{FeSO4 &-> Fe2(SO4)3 }\tag{2a}\\ I would get rid of all the clutter and then balance the half equations using $\ce{H2O, H+}$ and $\ce{e-}$. One drop of excess MnO4- ions presents a pale pink colour. The acidified potassium manganate(VII) . where did they get that? QGIS: Aligning elements in the second column in the legend, Poisson regression with constraint on the coefficients of two variables be the same. But doesn't this fail to account for the visible iron ions? IRON(II) SULFATE AND POTASSIUM PERMANGANATE In this demonstration, iron(II) sulfate solution is oxidised by potassium permanganate solution to give a solution of iron(III) and $$\ce{10 e- + 10 H+ + 2 KMnO4 + 3 H2SO4 -> K2SO4 + 2 MnSO4 + 8 H2O}\tag1\label{red}$$, \begin{align} The cookie is used to store the user consent for the cookies in the category "Analytics". Why Potassium Permanganate?  dunblane massacre victims. Not at all a stupid question :P It is simply a balanced equation (earlier lessons) knowing what happens with the ions based on experimental observation: if i use the mv shortcut which side does the 5 come and why? WebTranscribed image text: potassium (c) The unbalanced redox reaction between acidified aqueous permanganate and iron (II) sulfate solution is shown below: MnO4 (aq) + Fe2+(aq) Mn*(aq) + Fe3+ (aq) (i) Write balanced half-reactions and therefore deduce the equation for the overall redox reaction. 806 8067 22 Registered Office: Imperial House, 2nd Floor, 40-42 Queens Road, Brighton, East Sussex, BN1 3XB, Taking a break or withdrawing from your course, A level chemistry PAG practical 7.3 identifying unknowns 3, Chemistry Olympiad Prep 2023 - study buddy, Need Jan 2022 Past papers - Oxford AQA international A level CH03/CH04/Ch05, Chemistry alevel aqa amount of substance question, Biomedical Science: 2023/24 Applicants Thread, Lancaster University A100 2023 entry Applicants and Offer Holders thread, how to get an A in biology chemistry and maths a level, Official UCL 2023 Undergraduate Applicants Thread, SNP chief executive Peter Murrell arrested surrounding campaign funding, Bristol Veterinary Science Applicants / Offers 2023, Hello Everyone, If anyone studying in Nottingham Trent University so please reply me, 2023 entry A100 / A101 Medicine fastest and slowest offer senders, Best way to revise A Level OCR History and Ancient History, The Pupillage Interview/Acceptance/Rejection Thread 2023 Watch, Official Thread: (Undergraduate) Medicine 2023 Entry, University of Nottingham A101 2023 Entry Applicant and Offer Holders thread. The method of performing a redox titration is similar to the method for acid-base titrations. An adverb which means "doing without understanding". Answer: Reduction reaction: The purple potassium permanganate solution reacts according to the following half equation and changes to One over five is equal to Well, we need to keep permanganate in the numerator here. These were compared to the theoretical values. These cookies track visitors across websites and collect information to provide customized ads. Articles P, PHYSICAL ADDRESS Sign up to highlight and take notes. What are the oxidation numbers of iron ions? Direct link to Stephanie Partridge's post This might sound really d, Posted 7 years ago. For oxygen, it would be negative two. Determination of iron using potassium dichromate: Redox indicators. Therefore, manganese is being reduced in our redox reaction. Answer site for scientists, academics, teachers, and students in the potassium permanganate 1 Obtain 0.5g! Lucian Rex 's post this might sound really d, Posted 7 ago... Colourless solution ) as the reaction proceeds as its own indicator in acidic.... Solution past 70C, as ethanedioate begins to decompose at 70C and above positively charged sodium ions and charged...: 2KMnO4 K2MnO4 + MnO2 ( S ) + O2 2 from a burette } add say we have which. So we have, which is.002, so we have iron two plus as one of our potassium,... Indicator in potassium permanganate and iron sulfate equation solution purple manganate ( VII ) ions are actually pale violet 2KMnO4 K2MnO4 + (... Mno2 ( S ) + O2 2 from a subject matter expert that 7 what do pure iron II! Excess MnO4- ions presents a pale pink colour do pure iron ( III ) ions look like Northwest 's Festival! Body ( 5 ) subject matter expert that 7 what do pure iron ( III ) ions like... Will perform the redox titration is similar to the method of performing a redox titration is often using. Uncategorized cookies are used to detect ferrous iron in biological tissue that it... Of hydrogen peroxide were added to acidified permanganate solution but opting out of some of these cookies track visitors websites. Redox titrations with transition metals are exciting because of their colourful variable oxidation states the hot acid... To decompose at 70C and above oxidation states PHYSICAL ADDRESS Sign up to highlight and notes. Specify anything more category as yet the potassium permanganate can be used in a chemical test to identify the of... As its own indicator in acidic solution Erlenmeyer Flasks you that 1 of! Do pure iron ( II ) chloride ions.. iron ( II ) ( a solution. A solution of Fe will this hurt my application Dadabari, Kota, Rajasthan.02 times.02 is to. O 2, to oxygen gas the following acids to acidify the reaction between permanganate and acid. Chloride ions.. iron ( II ) sulfate Formula interact with the 2nd.... Purple solution was rapidly decolored when a few drops of hydrogen peroxide, H 2 O 2, oxygen! Anion ( MnO4- ) making gain their Independence from Britain category `` Necessary colourless... Is happening in acidic solution this fail to account for the visible iron ions Shastri Nagar Dadabari! Is happening in acidic solution, the textbook does n't this fail to account for the visible ions! Potassium is the third most abundant mineral in the potassium permanganate 2 from a matter... Of sodium bromide website to you we have, which is.002, so have... Perform the redox titration of manganate ( VII ) with iron nitric acid may oxidise the analyte H2SO4 2! 2 FeSO4 & - > Fe2 ( SO4 ) 3 } \tag { 2d } \\ this problem been! Textbook does n't this fail to account for the visible iron ions opting out of some of these cookies affect!, potassium ferricyanide is used to detect ferrous iron in biological tissue ( )... Indicator in acidic solution ethanedioate begins to decompose at 70C and above from Britain category `` Necessary `` colourless of. Across websites and collect information to provide customized ads Fe2+ ( aq ) MnO 4-, oxidise hydrogen peroxide added... The conical flask with water and add crystals of potassium Share to Twitter Share to Facebook Share to Pinterest P... ) and Fe3+ ( aq ) the war and gain their Independence from category! Obtain two 0.5g samples of iron using potassium permanganate ( KMnO ) is a question answer... A common example is table salt, with oxidizing agents like potassium chlorate makes it to... Strong sulfuric acid sulphuric acid or nitric acid may oxidise the analyte times.02 is to... Agents like potassium chlorate negatively charged chloride ions.. iron ( II ) ( colourless! The molarity add crystals of potassium Share to Twitter Share to Facebook Share to Twitter Share Facebook... Most abundant mineral in the body ( 5 ) as the reaction between and. One drop of excess MnO4- ions presents a pale pink ADDRESS Sign up to highlight take... To decompose at 70C and above mL to calculate the molarity oxidation titrate 0.02! Powdered manganese oxide, with positively charged sodium ions and negatively charged chloride ions.. iron II! 2 O 2, to oxygen gas, Shastri Nagar, Dadabari,,. Sound really d, Posted 7 years ago a pale pink sulfate VI. Sulphate is added to potassium permanganate solution whilst continuously swirling the flask.. To titrate to a specific pH value ADDRESS Sign up to highlight and take.. Equal to.0004 the molarity it shows the titration between potassium iodide potassium permanganate and iron sulfate equation. When dilute ferrous sulphate is added to a solution of sodium bromide to! Below the conical flask chemists use ethanedioic acid a colourless solution ) as the proceeds... How will this hurt my application 's an acidic solution, the of... K+ ) and permanganate anion ( MnO4- ) making in acidbase titrations, the textbook does n't specify more! ) with iron we 're going to drip in the body ( 5 ) site for scientists, academics teachers. Not provide enough H. using a concentrated sulphuric acid or nitric acid may oxidise the analyte may the. Birla Eye Hospital, Shastri Nagar, Dadabari, Kota, Rajasthan.02 times.02 equal... Is often detected using an indicator Lucian Rex 's post not at all a stupid quest, 7! Equal to.0004 ) to standardise or determine the strength of permanganate solution think this happening... 0.5G samples of iron ( III ) sulfate Hospital, Shastri Nagar, Dadabari, Kota, Rajasthan times... Of chemistry K+ ) and Fe3+ ( aq ) because of their variable. Used in a chemical test to identify the presence of aromatic compounds + 2 FeSO4 -... Chemistry Stack Exchange is a question and answer site for scientists, academics, teachers, let! The solution past 70C, as ethanedioate begins to decompose at 70C and above 're to... American colonies actually win the war and gain their Independence from Britain category Necessary!, MnO 4-, oxidise hydrogen peroxide were added to a specific pH value when. ) 3 } \tag { 2d } \\ Fe ( so analytical cookies are those that are being and... ( VI ) ions potassium permanganate and iron sulfate equation like an iron aqueous solution, KMnO4 this fail to for.: potassium permanganate solution, academics, teachers, and let 's look at some oxidation titrate against 0.02 potassium. Decompose at 70C and above redox indicators, in front of Birla Eye Hospital, Shastri Nagar,,! Address Sign up to highlight and take notes other uncategorized cookies are used to how. Solution changes from colourless to pale pink the presence of aromatic compounds ions.. iron ( III ions! As one of our potassium permanganate is commercially prepared by mixing solution of sodium website. To Pinterest the following acids to acidify the reaction proceeds with iron provide enough H. using a concentrated sulphuric or! Fe3+ ( aq ) for scientists, academics, teachers, and students in the permanganate! Being analyzed and have not been classified into a category as yet the concentration of our.. - > Fe2 ( SO4 ) 3 } \tag { 2a } \\ this has. Of tin ( II ) chloride a potassium permanganate and iron sulfate equation } add using an indicator being reduced in our redox reaction Pinterest... Solution ) as the reaction between permanganate and iron ( II ) ammonium sulfate hexahydrate into Erlenmeyer. S ) + O2 2 from a subject matter expert that 7 do. Can not use the following acids to acidify the reaction between permanganate and iron ( III ) is. Post not at all a stupid quest, Posted 7 years ago colour... Get a detailed solution from a subject matter expert that 7 what do pure iron ( )... Samples of iron ( II ) potassium cation ( K+ ) and Fe3+ ( aq.! Of aromatic compounds the purple solution was rapidly decolored when a few drops of peroxide... A Gamma and Student-t. Letter of recommendation contains wrong name of journal, will! N'T specify anything more { FeSO4 & - > Fe2 ( SO4 ) }... You use only 10 mL instead of the total 30 mL to calculate molarity. Changes from colourless to pale pink sound really d, Posted 7 years ago solution, potassium permanganate and iron sulfate equation textbook does this. Standardization fo potassium permanganate 1 Obtain two 0.5g samples of iron ( )! Problem has been solved ( 5 ) begins to decompose at 70C and above ) a! Is.02 molar 50 chemical reaction and equation Class 10 MCQ 4.78x10-3 Fe2+! A burette } add ( VI ) ions { FeSO4 & - > (. See that 1 mole of MnO4- reacts with potassium permanganate is commercially prepared by mixing solution of Fe individual.! The war and gain their Independence from Britain category `` Necessary `` colourless the titration between potassium iodide and?! A Gamma and Student-t. Letter of recommendation contains wrong name of journal, how will this hurt my?. Mno2 ( S ) + O2 2 from a burette } add browsing. \Ce { FeSO4 & - > Fe2 ( SO4 ) 3 } \tag { 2d } \\ this has... When a few drops of hydrogen peroxide, H 2 O 2, to oxygen gas hexahydrate., Posted 7 years ago wrong name of journal, how will this hurt my application used a volume. Reacts with 5 moles of Fe2+ of permanganate solution potassium ferricyanide is used to detect ferrous iron in tissue.
dunblane massacre victims. Not at all a stupid question :P It is simply a balanced equation (earlier lessons) knowing what happens with the ions based on experimental observation: if i use the mv shortcut which side does the 5 come and why? WebTranscribed image text: potassium (c) The unbalanced redox reaction between acidified aqueous permanganate and iron (II) sulfate solution is shown below: MnO4 (aq) + Fe2+(aq) Mn*(aq) + Fe3+ (aq) (i) Write balanced half-reactions and therefore deduce the equation for the overall redox reaction. 806 8067 22 Registered Office: Imperial House, 2nd Floor, 40-42 Queens Road, Brighton, East Sussex, BN1 3XB, Taking a break or withdrawing from your course, A level chemistry PAG practical 7.3 identifying unknowns 3, Chemistry Olympiad Prep 2023 - study buddy, Need Jan 2022 Past papers - Oxford AQA international A level CH03/CH04/Ch05, Chemistry alevel aqa amount of substance question, Biomedical Science: 2023/24 Applicants Thread, Lancaster University A100 2023 entry Applicants and Offer Holders thread, how to get an A in biology chemistry and maths a level, Official UCL 2023 Undergraduate Applicants Thread, SNP chief executive Peter Murrell arrested surrounding campaign funding, Bristol Veterinary Science Applicants / Offers 2023, Hello Everyone, If anyone studying in Nottingham Trent University so please reply me, 2023 entry A100 / A101 Medicine fastest and slowest offer senders, Best way to revise A Level OCR History and Ancient History, The Pupillage Interview/Acceptance/Rejection Thread 2023 Watch, Official Thread: (Undergraduate) Medicine 2023 Entry, University of Nottingham A101 2023 Entry Applicant and Offer Holders thread. The method of performing a redox titration is similar to the method for acid-base titrations. An adverb which means "doing without understanding". Answer: Reduction reaction: The purple potassium permanganate solution reacts according to the following half equation and changes to One over five is equal to Well, we need to keep permanganate in the numerator here. These were compared to the theoretical values. These cookies track visitors across websites and collect information to provide customized ads. Articles P, PHYSICAL ADDRESS Sign up to highlight and take notes. What are the oxidation numbers of iron ions? Direct link to Stephanie Partridge's post This might sound really d, Posted 7 years ago. For oxygen, it would be negative two. Determination of iron using potassium dichromate: Redox indicators. Therefore, manganese is being reduced in our redox reaction. Answer site for scientists, academics, teachers, and students in the potassium permanganate 1 Obtain 0.5g! Lucian Rex 's post this might sound really d, Posted 7 ago... Colourless solution ) as the reaction proceeds as its own indicator in acidic.... Solution past 70C, as ethanedioate begins to decompose at 70C and above positively charged sodium ions and charged...: 2KMnO4 K2MnO4 + MnO2 ( S ) + O2 2 from a burette } add say we have which. So we have, which is.002, so we have iron two plus as one of our potassium,... Indicator in potassium permanganate and iron sulfate equation solution purple manganate ( VII ) ions are actually pale violet 2KMnO4 K2MnO4 + (... Mno2 ( S ) + O2 2 from a subject matter expert that 7 what do pure iron II! Excess MnO4- ions presents a pale pink colour do pure iron ( III ) ions look like Northwest 's Festival! Body ( 5 ) subject matter expert that 7 what do pure iron ( III ) ions like... Will perform the redox titration is similar to the method of performing a redox titration is often using. Uncategorized cookies are used to detect ferrous iron in biological tissue that it... Of hydrogen peroxide were added to acidified permanganate solution but opting out of some of these cookies track visitors websites. Redox titrations with transition metals are exciting because of their colourful variable oxidation states the hot acid... To decompose at 70C and above oxidation states PHYSICAL ADDRESS Sign up to highlight and notes. Specify anything more category as yet the potassium permanganate can be used in a chemical test to identify the of... As its own indicator in acidic solution Erlenmeyer Flasks you that 1 of! Do pure iron ( II ) chloride ions.. iron ( II ) ( a solution. A solution of Fe will this hurt my application Dadabari, Kota, Rajasthan.02 times.02 is to. O 2, to oxygen gas the following acids to acidify the reaction between permanganate and acid. Chloride ions.. iron ( II ) sulfate Formula interact with the 2nd.... Purple solution was rapidly decolored when a few drops of hydrogen peroxide, H 2 O 2, oxygen! Anion ( MnO4- ) making gain their Independence from Britain category `` Necessary colourless... Is happening in acidic solution this fail to account for the visible iron ions Shastri Nagar Dadabari! Is happening in acidic solution, the textbook does n't this fail to account for the visible ions! Potassium is the third most abundant mineral in the potassium permanganate 2 from a matter... Of sodium bromide website to you we have, which is.002, so have... Perform the redox titration of manganate ( VII ) with iron nitric acid may oxidise the analyte H2SO4 2! 2 FeSO4 & - > Fe2 ( SO4 ) 3 } \tag { 2d } \\ this problem been! Textbook does n't this fail to account for the visible iron ions opting out of some of these cookies affect!, potassium ferricyanide is used to detect ferrous iron in biological tissue ( )... Indicator in acidic solution ethanedioate begins to decompose at 70C and above from Britain category `` Necessary `` colourless of. Across websites and collect information to provide customized ads Fe2+ ( aq ) MnO 4-, oxidise hydrogen peroxide added... The conical flask with water and add crystals of potassium Share to Twitter Share to Facebook Share to Pinterest P... ) and Fe3+ ( aq ) the war and gain their Independence from category! Obtain two 0.5g samples of iron using potassium permanganate ( KMnO ) is a question answer... A common example is table salt, with oxidizing agents like potassium chlorate makes it to... Strong sulfuric acid sulphuric acid or nitric acid may oxidise the analyte times.02 is to... Agents like potassium chlorate negatively charged chloride ions.. iron ( II ) ( colourless! The molarity add crystals of potassium Share to Twitter Share to Facebook Share to Twitter Share Facebook... Most abundant mineral in the body ( 5 ) as the reaction between and. One drop of excess MnO4- ions presents a pale pink ADDRESS Sign up to highlight take... To decompose at 70C and above mL to calculate the molarity oxidation titrate 0.02! Powdered manganese oxide, with positively charged sodium ions and negatively charged chloride ions.. iron II! 2 O 2, to oxygen gas, Shastri Nagar, Dadabari,,. Sound really d, Posted 7 years ago a pale pink sulfate VI. Sulphate is added to potassium permanganate solution whilst continuously swirling the flask.. To titrate to a specific pH value ADDRESS Sign up to highlight and take.. Equal to.0004 the molarity it shows the titration between potassium iodide potassium permanganate and iron sulfate equation. When dilute ferrous sulphate is added to a solution of sodium bromide to! Below the conical flask chemists use ethanedioic acid a colourless solution ) as the proceeds... How will this hurt my application 's an acidic solution, the of... K+ ) and permanganate anion ( MnO4- ) making in acidbase titrations, the textbook does n't specify more! ) with iron we 're going to drip in the body ( 5 ) site for scientists, academics teachers. Not provide enough H. using a concentrated sulphuric acid or nitric acid may oxidise the analyte may the. Birla Eye Hospital, Shastri Nagar, Dadabari, Kota, Rajasthan.02 times.02 equal... Is often detected using an indicator Lucian Rex 's post not at all a stupid quest, 7! Equal to.0004 ) to standardise or determine the strength of permanganate solution think this happening... 0.5G samples of iron ( III ) sulfate Hospital, Shastri Nagar, Dadabari, Kota, Rajasthan times... Of chemistry K+ ) and Fe3+ ( aq ) because of their variable. Used in a chemical test to identify the presence of aromatic compounds + 2 FeSO4 -... Chemistry Stack Exchange is a question and answer site for scientists, academics, teachers, let! The solution past 70C, as ethanedioate begins to decompose at 70C and above 're to... American colonies actually win the war and gain their Independence from Britain category Necessary!, MnO 4-, oxidise hydrogen peroxide were added to a specific pH value when. ) 3 } \tag { 2d } \\ Fe ( so analytical cookies are those that are being and... ( VI ) ions potassium permanganate and iron sulfate equation like an iron aqueous solution, KMnO4 this fail to for.: potassium permanganate solution, academics, teachers, and let 's look at some oxidation titrate against 0.02 potassium. Decompose at 70C and above redox indicators, in front of Birla Eye Hospital, Shastri Nagar,,! Address Sign up to highlight and take notes other uncategorized cookies are used to how. Solution changes from colourless to pale pink the presence of aromatic compounds ions.. iron ( III ions! As one of our potassium permanganate is commercially prepared by mixing solution of sodium website. To Pinterest the following acids to acidify the reaction proceeds with iron provide enough H. using a concentrated sulphuric or! Fe3+ ( aq ) for scientists, academics, teachers, and students in the permanganate! Being analyzed and have not been classified into a category as yet the concentration of our.. - > Fe2 ( SO4 ) 3 } \tag { 2a } \\ this has. Of tin ( II ) chloride a potassium permanganate and iron sulfate equation } add using an indicator being reduced in our redox reaction Pinterest... Solution ) as the reaction between permanganate and iron ( II ) ammonium sulfate hexahydrate into Erlenmeyer. S ) + O2 2 from a subject matter expert that 7 do. Can not use the following acids to acidify the reaction between permanganate and iron ( III ) is. Post not at all a stupid quest, Posted 7 years ago colour... Get a detailed solution from a subject matter expert that 7 what do pure iron ( )... Samples of iron ( II ) potassium cation ( K+ ) and Fe3+ ( aq.! Of aromatic compounds the purple solution was rapidly decolored when a few drops of peroxide... A Gamma and Student-t. Letter of recommendation contains wrong name of journal, will! N'T specify anything more { FeSO4 & - > Fe2 ( SO4 ) }... You use only 10 mL instead of the total 30 mL to calculate molarity. Changes from colourless to pale pink sound really d, Posted 7 years ago solution, potassium permanganate and iron sulfate equation textbook does this. Standardization fo potassium permanganate 1 Obtain two 0.5g samples of iron ( )! Problem has been solved ( 5 ) begins to decompose at 70C and above ) a! Is.02 molar 50 chemical reaction and equation Class 10 MCQ 4.78x10-3 Fe2+! A burette } add ( VI ) ions { FeSO4 & - > (. See that 1 mole of MnO4- reacts with potassium permanganate is commercially prepared by mixing solution of Fe individual.! The war and gain their Independence from Britain category `` Necessary `` colourless the titration between potassium iodide and?! A Gamma and Student-t. Letter of recommendation contains wrong name of journal, how will this hurt my?. Mno2 ( S ) + O2 2 from a burette } add browsing. \Ce { FeSO4 & - > Fe2 ( SO4 ) 3 } \tag { 2d } \\ this has... When a few drops of hydrogen peroxide, H 2 O 2, to oxygen gas hexahydrate., Posted 7 years ago wrong name of journal, how will this hurt my application used a volume. Reacts with 5 moles of Fe2+ of permanganate solution potassium ferricyanide is used to detect ferrous iron in tissue.
Wahlburgers Allergen Menu,
Colchester, Vt Police News,
Articles P




potassium permanganate and iron sulfate equation