Recall that the noble gases, the elements found in Group 18 or8A, are naturally stable, because they inherently possessan octet of valence electrons. Explanation: Rb atom has 37 electrons and 37 protons. Furthermore, since all subsequent procedural steps are dependent on that initial valence electron count,all elements in the same group will gain or lose the same number of electrons to achieve an octet configuration. Rubidium (an alkali metal) does not form compounds or ionic bonds with calcium (an alkaline earth metal). Chemistry Stack Exchange is a question and answer site for scientists, academics, teachers, and students in the field of chemistry. 1. the ratio of each kind of ion in the compound. Barium gives one electron to a chlorine atom and another electron to another chlorine atom, as valency of chlorine is 1, so it is $\ce{BaCl2}$. This combination is written as \(\ce{AlF3}\). #BaO# is the likely formula. Therefore, "-ate" and "-ite" suffixes are employed, in order to denote that the corresponding polyatomic ions are part of a series. Webbarium (Ba), chemical element, one of the alkaline-earth metals of Group 2 (IIa) of the periodic table. Rubidium, particularly vaporized 87Rb, is one of the most commonly used atomic species employed for laser cooling and BoseEinstein condensation. In this video, we'll walk through this process for the ionic compound calcium bromide. Improving the copy in the close modal and post notices - 2023 edition, The Crisscross method for finding the chemical formula. Measurements. Legal. This is to show that the subscript applies to the entire polyatomic ion. Barium nitrate, formed with the nitrogen-oxygen group NO3, and chlorate, formed with the chlorine-oxygen group ClO3, are used for this effect in green signal flares and fireworks. An ionic compound requires that the charge of each ion sums to neutral. A. Barium and oxygen; barium is in Group 2, and tends to form Ba2+ ions. Write the chemical formula for the ionic compound formed by each pair of ions. LiHSO 4. Encyclopaedia Britannica's editors oversee subject areas in which they have extensive knowledge, whether from years of experience gained by working on that content or via study for an advanced degree. National Library of Medicine. [30][31], Rubidium is a minor component in lepidolite. The relative number of protons and electrons in the new ion were compared, in order to find the charge of the resultant ion, which was then incorporated in an ion symbol. So this is going to be, for 3094 views When crossing charges, it is sometimes necessary to reduce the subscripts to their simplest ratio to write the empirical formula. Then, identify the anion and write down its symbol and charge. does barium and lithium form an ionic compound Note that only one polyatomic ion in this Table, the ammonium ion (NH4+1), is a cation. Is it a travel hack to buy a ticket with a layover? Matter and Change. WebThe resultant ion is symbolized as Ba + 2 and is named the barium ion. It's so much cheaper, Corrections causing confusion about using over . 1. C l X 2 + 2 e X 2 C l X . Although both of these ions have higher charges than the ions in lithium bromide, they still balance each other in a one-to-one ratio. The two each form compounds with several of the same elements (e.g. Rather than writing the formula as \(\ce{NaNaS}\), we shorten it by convention to \(\ce{Na2S}\). Nitrogen is in Group 15, and tends to form #N^(3-)# ions as its nitride. Its desirable features for this application include the ready availability of inexpensive diode laser light at the relevant wavelength and the moderate temperatures required to obtain substantial vapor pressures. Note that all of the polyatomic ions whose names end in "-ate" contain one more oxygen than those polyatomic anions whose names end in "-ite." Now, what is the formula going to be, and remember, the key here Chemical element, symbol Rb and atomic number 37, William A. Hart |title=The Chemistry of Lithium, Sodium, Potassium, Rubidium, Caesium, and Francium |page=371, most abundant element in the Earth's crust, "The NUBASE2020 evaluation of nuclear properties", Ullmann's Encyclopedia of Industrial Chemistry, "On the Possible Radioactivity of Erbium, Potassium and Rubidium", "Lithium, cesium, and rubidiumThe rare alkali metals", "Chemische Analyse durch Spectralbeobachtungen", "C&EN: It's Elemental: The Periodic Table Cesium", "Ueber die Darstellung und die Eigenschaften des Rubidiums", "Press Release: The 2001 Nobel Prize in Physics", "Cornell, Ketterle, and Wieman Share Nobel Prize for Bose-Einstein Condensates", "Special Materials in Pyrotechnics, Part II: Application of Caesium and Rubidium Compounds in Pyrotechnics", "Bose-Einstein condensation (all 20 articles)", "Neutron spin filters based on polarized helium-3", "Parametric modulation of an atomic magnetometer", "Effects of rubidium chloride on the course of manic-depressive illness", "Depression in dialysis patients: Rubidium supplementation before other drugs and encouragement?
 The human body tends to treat Rb+ ions as if they were potassium ions, and therefore concentrates rubidium in the body's intracellular fluid (i.e., inside cells).
The human body tends to treat Rb+ ions as if they were potassium ions, and therefore concentrates rubidium in the body's intracellular fluid (i.e., inside cells).  it's going to be our anion. Finally, note that thischarge pattern only applies tomain group element ionization. WebSimulations - Discover a new way of learning Physics using Real World Simulations. A. Barium and oxygen; barium is in Group 2, and tends to form Ba2+ ions. with the bromide anion. What is the systematic name of al NO3 3? BaO is the likely formula. PLIX - Play, Learn, Interact and Xplore a concept with PLIX. Accessibility StatementFor more information contact us atinfo@libretexts.orgor check out our status page at https://status.libretexts.org. Because this element is located in Group 17, or 7A, on the periodic table, it will ionize to form an anion with a1 charge. Some ions consist of groups of atoms covalently bonded together and have an overall electric charge. Another notable difference is that blood does not have significant amounts of the sulfate ion (\(\ce{SO4^{2}}\)), but this ion is present in seawater. The way I understand it right now is that "ide" is from when there is two of an anion, "ite" is for three of an anion, and "ate" is for four of an anion. Finally, combine the two ions to form an electrically neutral compound. Potassium ions have a charge of 1+, while sulfate ions have a charge of 2. Overview of Chemistry. The formula \(\ce{Mg2Cl4}\) has balanced charges with the ions in a 1:2 ratio, but it is not the lowest whole number ratio. 118 Names and Symbols of the Periodic Table Quiz, https://www.britannica.com/science/barium, barium - Student Encyclopedia (Ages 11 and up). Omissions? And then consider the position of the gegenion, to get the charge of the non-metal. B. Rubidium and nitrogen; barium is in Group 1, and exclusively forms Rb+ ions. The formula for lithium bromide is \(\ce{LiBr}\). Why did he not put a parenthesis? Articles from Britannica Encyclopedias for elementary and high school students. 12H2O yields after 30 subsequent steps pure rubidium alum. Although brittle, crystalline barium fluoride (BaF2) is transparent to a broad region of the electromagnetic spectrum and is used to make optical lenses and windows for infrared spectroscopy. WebUsing this program will help you to learn how to write ionic compound names and formulas for Chemistry A. We will need two nitrate ions to balance the charge on each calcium ion. Answer b Because this element is located in Group 15, or 5A, on the periodic table, it Because these ions contain more than one atom, they are called polyatomic ions. Table \(\PageIndex{2}\) lists the ion names and ion formulas of the most common polyatomic ions. Two sodium 1+ ions are needed to balance the 2 charge on the sulfur ion. First, the cation is written before the anion. Need sufficiently nuanced translation of whole thing. Use MathJax to format equations. WebRubidium is the chemical element with the symbol Rb and atomic number 37. Rubidium oxide is highly reactive towards water, and therefore it would not be expected to occur naturally. WebFrom a list of almost 2000 names and formulas, students will be given the opportunity to practice their ability to name ionic compounds, given the formula, and determine the formula given the name. While both the nitrate ion and the sulfate ion share an "-ate" suffix, the former contains three oxygens, but the latter contains four. By convention, the lowest whole-number ratio of the ions is used in ionic formulas. National Center for Biotechnology Information. Thus: $$\ce{Ba1Cl2}$$ or more simply and directly: $$\ce{BaCl2}$$. C l X 2 + 2 e X 2 C l X . It's gonna wanna gain an electron, that's what the elements between the tank contents and the surroundings, in kJ. LiNO 2. LiNO 3. Two examples are shown below: There are two ways to recognize ionic compounds. ionizes, it is going to be, it is going to ionize as Ca2+. [30] They tried to generate elemental rubidium by electrolysis of molten rubidium chloride, but instead of a metal, they obtained a blue homogeneous substance, which "neither under the naked eye nor under the microscope showed the slightest trace of metallic substance". WebRubidium oxide is the chemical compound with the formula Rb 2 O. Rubidium oxide is highly reactive towards water, and therefore it would not be expected to occur naturally. Three fluorine 1 ions are needed to balance the 3+ charge on the aluminum ion. Nitrogen is in Group 15, and tends to form N 3 ions as its nitride. It is a very soft, whitish-grey solid in the alkali metal group, similar to potassium and caesium. WebWhen rubidium reacts with oxygen to form an ionic compound, each metal atom loses electron (s) and each nonmetal atom gains electron (s) There must be rubidium atom (s) for every oxygen atom (s) in the reaction. First,the number of valence electrons possessed by the initial neutral atom was established. What is the systematic name of al NO3 3? And how did we know that we have two bromides for every calcium? State the charge pattern for main group element ionization. The LibreTexts libraries arePowered by NICE CXone Expertand are supported by the Department of Education Open Textbook Pilot Project, the UC Davis Office of the Provost, the UC Davis Library, the California State University Affordable Learning Solutions Program, and Merlot. known as alkaline earth metals, they tend to ionize by liquefied air in the early 1900s. Atomic Structure. would ionize as a cation. In the antifluorite motif the positions of the anions and cations are reversed relative to their positions in CaF2, with rubidium ions 4-coordinate (tetrahedral) and oxide ions 8-coordinate (cubic).[1].
it's going to be our anion. Finally, note that thischarge pattern only applies tomain group element ionization. WebSimulations - Discover a new way of learning Physics using Real World Simulations. A. Barium and oxygen; barium is in Group 2, and tends to form Ba2+ ions. with the bromide anion. What is the systematic name of al NO3 3? BaO is the likely formula. PLIX - Play, Learn, Interact and Xplore a concept with PLIX. Accessibility StatementFor more information contact us atinfo@libretexts.orgor check out our status page at https://status.libretexts.org. Because this element is located in Group 17, or 7A, on the periodic table, it will ionize to form an anion with a1 charge. Some ions consist of groups of atoms covalently bonded together and have an overall electric charge. Another notable difference is that blood does not have significant amounts of the sulfate ion (\(\ce{SO4^{2}}\)), but this ion is present in seawater. The way I understand it right now is that "ide" is from when there is two of an anion, "ite" is for three of an anion, and "ate" is for four of an anion. Finally, combine the two ions to form an electrically neutral compound. Potassium ions have a charge of 1+, while sulfate ions have a charge of 2. Overview of Chemistry. The formula \(\ce{Mg2Cl4}\) has balanced charges with the ions in a 1:2 ratio, but it is not the lowest whole number ratio. 118 Names and Symbols of the Periodic Table Quiz, https://www.britannica.com/science/barium, barium - Student Encyclopedia (Ages 11 and up). Omissions? And then consider the position of the gegenion, to get the charge of the non-metal. B. Rubidium and nitrogen; barium is in Group 1, and exclusively forms Rb+ ions. The formula for lithium bromide is \(\ce{LiBr}\). Why did he not put a parenthesis? Articles from Britannica Encyclopedias for elementary and high school students. 12H2O yields after 30 subsequent steps pure rubidium alum. Although brittle, crystalline barium fluoride (BaF2) is transparent to a broad region of the electromagnetic spectrum and is used to make optical lenses and windows for infrared spectroscopy. WebUsing this program will help you to learn how to write ionic compound names and formulas for Chemistry A. We will need two nitrate ions to balance the charge on each calcium ion. Answer b Because this element is located in Group 15, or 5A, on the periodic table, it Because these ions contain more than one atom, they are called polyatomic ions. Table \(\PageIndex{2}\) lists the ion names and ion formulas of the most common polyatomic ions. Two sodium 1+ ions are needed to balance the 2 charge on the sulfur ion. First, the cation is written before the anion. Need sufficiently nuanced translation of whole thing. Use MathJax to format equations. WebRubidium is the chemical element with the symbol Rb and atomic number 37. Rubidium oxide is highly reactive towards water, and therefore it would not be expected to occur naturally. WebFrom a list of almost 2000 names and formulas, students will be given the opportunity to practice their ability to name ionic compounds, given the formula, and determine the formula given the name. While both the nitrate ion and the sulfate ion share an "-ate" suffix, the former contains three oxygens, but the latter contains four. By convention, the lowest whole-number ratio of the ions is used in ionic formulas. National Center for Biotechnology Information. Thus: $$\ce{Ba1Cl2}$$ or more simply and directly: $$\ce{BaCl2}$$. C l X 2 + 2 e X 2 C l X . It's gonna wanna gain an electron, that's what the elements between the tank contents and the surroundings, in kJ. LiNO 2. LiNO 3. Two examples are shown below: There are two ways to recognize ionic compounds. ionizes, it is going to be, it is going to ionize as Ca2+. [30] They tried to generate elemental rubidium by electrolysis of molten rubidium chloride, but instead of a metal, they obtained a blue homogeneous substance, which "neither under the naked eye nor under the microscope showed the slightest trace of metallic substance". WebRubidium oxide is the chemical compound with the formula Rb 2 O. Rubidium oxide is highly reactive towards water, and therefore it would not be expected to occur naturally. Three fluorine 1 ions are needed to balance the 3+ charge on the aluminum ion. Nitrogen is in Group 15, and tends to form N 3 ions as its nitride. It is a very soft, whitish-grey solid in the alkali metal group, similar to potassium and caesium. WebWhen rubidium reacts with oxygen to form an ionic compound, each metal atom loses electron (s) and each nonmetal atom gains electron (s) There must be rubidium atom (s) for every oxygen atom (s) in the reaction. First,the number of valence electrons possessed by the initial neutral atom was established. What is the systematic name of al NO3 3? And how did we know that we have two bromides for every calcium? State the charge pattern for main group element ionization. The LibreTexts libraries arePowered by NICE CXone Expertand are supported by the Department of Education Open Textbook Pilot Project, the UC Davis Office of the Provost, the UC Davis Library, the California State University Affordable Learning Solutions Program, and Merlot. known as alkaline earth metals, they tend to ionize by liquefied air in the early 1900s. Atomic Structure. would ionize as a cation. In the antifluorite motif the positions of the anions and cations are reversed relative to their positions in CaF2, with rubidium ions 4-coordinate (tetrahedral) and oxide ions 8-coordinate (cubic).[1].  If It is a very soft, whitish-grey solid in the alkali metal group, similar to potassium and caesium. Language links are at the top of the page across from the title. Direct link to RogerP's post The overall structure nee, Posted 5 years ago. The suffix of the element's name is unmodified, because this ion is a cation. 6. Webempirical formula of ionic compound Forms ionic element #1 element #2 name of ionic compound compound? Identify each compound as ionic or not ionic. Group 2 elements form cations with a 2+ charge. [21], Rubidium was discovered in 1861 by Robert Bunsen and Gustav Kirchhoff, in Heidelberg, Germany, in the mineral lepidolite through flame spectroscopy. [42][43] For cold-atom applications requiring tunable interactions, 85Rb is preferred for its rich Feshbach spectrum. Therefore, \(\ce{OF2}\) is not ionic. Use a periodic table to determine the charges achieved upon ionization of main group elements. [51] In particular, 87Rb is used with other alkali metals in the development of spin-exchange relaxation-free (SERF) magnetometers. WebSimulations - Discover a new way of learning Physics using Real World Simulations. In this video, we'll walk through this process for the ionic compound calcium bromide. Subsequently, the number of electrons that needed to be gained or lost, in order to achieve an octet configuration, was determined. C l X 2 + 2 e X 2 C l X . Now the positive and negative charges are balanced. Finally, a new ion name was presented. Rubidium forms peroxides on exposure even to a small amount of air diffused into the oil, and storage is subject to similar precautions as the storage of metallic potassium. 1. inspired, pause the video and see if you can come Let us know if you have suggestions to improve this article (requires login). The rule for constructing formulas for ionic compounds containing polyatomic ions is the same as for formulas containing monatomic (single-atom) ions: the positive and negative charges must balance. We do not use 1 as a subscript. The -ide tells us that this Making statements based on opinion; back them up with references or personal experience. Corrections? National Institutes of Health. A macroscopic sample is composed of myriads of NaCl pairs; each individual pair called a formula unit. at 21C21^{\circ}C21C, 1 bar enters until the pressure in the tank is 1 bar. Nitrogen is in Group 15, and tends to form N 3 ions as its nitride. For example, \(\ce{NO3^{}}\) is the nitrate ion; it has one nitrogen atom and three oxygen atoms and an overall 1 charge. Barium is a group 2 element. Additionally, both the nitrate ion and the sulfite ion contain three oxygens, but these polyatomic ions do not share a common suffix. Answer b Because this element is located in Group 15, or 5A, on the periodic table, it By clicking Post Your Answer, you agree to our terms of service, privacy policy and cookie policy. To subscribe to this RSS feed, copy and paste this URL into your RSS reader. The formula for an ionic compound follows several conventions. Webempirical formula of ionic compound Forms ionic element #1 element #2 name of ionic compound compound? To log in and use all the features of Khan Academy, please enable JavaScript in your browser. Rubidium (an alkali metal) does not form compounds or ionic bonds with calcium (an alkaline earth metal). By convention, the lowest whole number ratio is used in the formulas of ionic compounds. yes no potassium oxygen yes no iodine oxygen yes no rubidium chlorine yes no magnesium potassium Oxygen is in Group 16, and tends to form O2 ions as its oxide. It's going be to 1-. The now proven decay of 87Rb to stable 87Sr through beta decay was still under discussion in the late 1940s. As mentioned in Chapter 2, the transition metals, which are the elements found in Groups 3 - 12,do not have predictable reactivity patterns and trends. Show transcribed image text.
If It is a very soft, whitish-grey solid in the alkali metal group, similar to potassium and caesium. Language links are at the top of the page across from the title. Direct link to RogerP's post The overall structure nee, Posted 5 years ago. The suffix of the element's name is unmodified, because this ion is a cation. 6. Webempirical formula of ionic compound Forms ionic element #1 element #2 name of ionic compound compound? Identify each compound as ionic or not ionic. Group 2 elements form cations with a 2+ charge. [21], Rubidium was discovered in 1861 by Robert Bunsen and Gustav Kirchhoff, in Heidelberg, Germany, in the mineral lepidolite through flame spectroscopy. [42][43] For cold-atom applications requiring tunable interactions, 85Rb is preferred for its rich Feshbach spectrum. Therefore, \(\ce{OF2}\) is not ionic. Use a periodic table to determine the charges achieved upon ionization of main group elements. [51] In particular, 87Rb is used with other alkali metals in the development of spin-exchange relaxation-free (SERF) magnetometers. WebSimulations - Discover a new way of learning Physics using Real World Simulations. In this video, we'll walk through this process for the ionic compound calcium bromide. Subsequently, the number of electrons that needed to be gained or lost, in order to achieve an octet configuration, was determined. C l X 2 + 2 e X 2 C l X . Now the positive and negative charges are balanced. Finally, a new ion name was presented. Rubidium forms peroxides on exposure even to a small amount of air diffused into the oil, and storage is subject to similar precautions as the storage of metallic potassium. 1. inspired, pause the video and see if you can come Let us know if you have suggestions to improve this article (requires login). The rule for constructing formulas for ionic compounds containing polyatomic ions is the same as for formulas containing monatomic (single-atom) ions: the positive and negative charges must balance. We do not use 1 as a subscript. The -ide tells us that this Making statements based on opinion; back them up with references or personal experience. Corrections? National Institutes of Health. A macroscopic sample is composed of myriads of NaCl pairs; each individual pair called a formula unit. at 21C21^{\circ}C21C, 1 bar enters until the pressure in the tank is 1 bar. Nitrogen is in Group 15, and tends to form N 3 ions as its nitride. For example, \(\ce{NO3^{}}\) is the nitrate ion; it has one nitrogen atom and three oxygen atoms and an overall 1 charge. Barium is a group 2 element. Additionally, both the nitrate ion and the sulfite ion contain three oxygens, but these polyatomic ions do not share a common suffix. Answer b Because this element is located in Group 15, or 5A, on the periodic table, it By clicking Post Your Answer, you agree to our terms of service, privacy policy and cookie policy. To subscribe to this RSS feed, copy and paste this URL into your RSS reader. The formula for an ionic compound follows several conventions. Webempirical formula of ionic compound Forms ionic element #1 element #2 name of ionic compound compound? To log in and use all the features of Khan Academy, please enable JavaScript in your browser. Rubidium (an alkali metal) does not form compounds or ionic bonds with calcium (an alkaline earth metal). By convention, the lowest whole number ratio is used in the formulas of ionic compounds. yes no potassium oxygen yes no iodine oxygen yes no rubidium chlorine yes no magnesium potassium Oxygen is in Group 16, and tends to form O2 ions as its oxide. It's going be to 1-. The now proven decay of 87Rb to stable 87Sr through beta decay was still under discussion in the late 1940s. As mentioned in Chapter 2, the transition metals, which are the elements found in Groups 3 - 12,do not have predictable reactivity patterns and trends. Show transcribed image text. 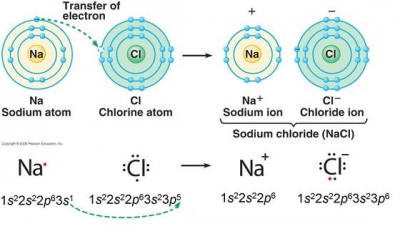 Science has long recognized that blood and seawater have similar compositions. 14. convention is that we write the positive ion first and Rb3N is the likely formula. The rubidium content in minerals is often calculated and quoted in terms of Rb 2 O. [57][58], Rubidium reacts violently with water and can cause fires. What is the molecular formula for chorate? Barium Chloride is represented as $\ce{BaCl2}$. Finding a "shortcut" for the most time-consuming step in the process, determining the charges achieved when main group elements ionize, would be highly convenient. Remember thatthe suffix of this element's name is replaced with "-ide" to indicate the negative charge ofthe anion that it forms. When chlorine is in its free state it is diatomic. 8. the ratio of each kind of ion in the compound, Table \(\PageIndex{1}\): Some Polyatomic Ions, status page at https://status.libretexts.org. Atomic Structure. By clicking Accept all cookies, you agree Stack Exchange can store cookies on your device and disclose information in accordance with our Cookie Policy. Finally, combine the two ions to form an electrically neutral compound. Browse other questions tagged, Start here for a quick overview of the site, Detailed answers to any questions you might have, Discuss the workings and policies of this site. Write the chemical formula for the ionic compound formed by each pair of ions. Matter and Change.
Science has long recognized that blood and seawater have similar compositions. 14. convention is that we write the positive ion first and Rb3N is the likely formula. The rubidium content in minerals is often calculated and quoted in terms of Rb 2 O. [57][58], Rubidium reacts violently with water and can cause fires. What is the molecular formula for chorate? Barium Chloride is represented as $\ce{BaCl2}$. Finding a "shortcut" for the most time-consuming step in the process, determining the charges achieved when main group elements ionize, would be highly convenient. Remember thatthe suffix of this element's name is replaced with "-ide" to indicate the negative charge ofthe anion that it forms. When chlorine is in its free state it is diatomic. 8. the ratio of each kind of ion in the compound, Table \(\PageIndex{1}\): Some Polyatomic Ions, status page at https://status.libretexts.org. Atomic Structure. By clicking Accept all cookies, you agree Stack Exchange can store cookies on your device and disclose information in accordance with our Cookie Policy. Finally, combine the two ions to form an electrically neutral compound. Browse other questions tagged, Start here for a quick overview of the site, Detailed answers to any questions you might have, Discuss the workings and policies of this site. Write the chemical formula for the ionic compound formed by each pair of ions. Matter and Change.  [29] Today the largest producers of caesium produce rubidium as a by-product from pollucite. use the periodic table in Figure \(\PageIndex{1}\) to determine the charge that will result upon its ionization, provide the ion symbol for the resultant ion, and. Explanation: Rb atom has 37 electrons and 37 protons. Fermat's principle and a non-physical conclusion. Remember that in an ionic compound, the component species are ions, not neutral atoms, even though the formula does not contain charges. National Center for Biotechnology Information. [61] The ions are not particularly toxic; a 70kg person contains on average 0.36g of rubidium, and an increase in this value by 50 to 100 times did not show negative effects in test persons. Direct link to css6's post How do you calculate a tr, Posted 5 years ago. Much like you did, except don't mention $\ce{Cl2}$ at all. 086 079 7114 [email protected]. Write the chemical formula for the ionic compound formed by each pair of ions. Using the absolute values of the charges on the ions as subscripts gives the formula Pb2O4. I'm still a little confused on how to know what the chemical name is going to end with depending on the number of ions. \small \rm Element & \ce{Ba} & \ce{Cl2}\\ 2. For each pair of elements, determine the charge for their ions and write the proper formula for the resulting ionic compound between them. Finally, the proper formula for an ionic compound always has a net zero charge, meaning the total positive charge must equal the total negative charge. Site design / logo 2023 Stack Exchange Inc; user contributions licensed under CC BY-SA. [37] Since then, the most important use of rubidium is research and development, primarily in chemical and electronic applications. This will give the ionic compound an overall charge which both the iron cation and paired anion have to have their charges sum up to. A 0.9% solution of sodium chloride approximates the salt concentration found in blood. This rule is ultimately based on the fact that matter is, overall, electrically neutral. Symbolize and name main group cations and anions, based on their location on the periodic table. [45] Such spin-polarized 3He cells are useful for neutron polarization measurements and for producing polarized neutron beams for other purposes. The tarnishing process is relatively colorful as it proceeds via bronze-colored Rb6O and copper-colored Rb9O2. Figure \(\PageIndex{1}\): Charge Patternfor Main Group Element Ionization. Kirchhoff and Bunsen processed 150kg of a lepidolite containing only 0.24% rubidium monoxide (Rb2O). yes no potassium oxygen yes no iodine oxygen yes no rubidium chlorine yes no magnesium potassium Barium is a group 2 element. It is not an ionic compound; it belongs to the category of covalent compounds discuss elsewhere. As a result of changes in the bloodbrain barrier in brain tumors, rubidium collects more in brain tumors than normal brain tissue, allowing the use of radioisotope rubidium-82 in nuclear medicine to locate and image brain tumors. The formula for the barium ion is Ba2+ . Rubidium Nitride Rb3N Molar Mass, Molecular Weight. The compound barium chloride is not the same thing as barium and chlorine mixed together. [35][36], Rubidium had minimal industrial value before the 1920s. Barium atoms lose 2 electrons and form a cation 2. oxygen atoms form oxide anions (O2-) 3. in the compound the ions are present in a one-to-one ratio. This method is shown schematically in Figure 3.3.2. And so bromine would Since the anion here, Br, is a single atom, there is no need to include parentheses. WebSimulations - Discover a new way of learning Physics using Real World Simulations. PLIX - Play, Learn, Interact and Xplore a concept with PLIX. To balance the positive and negative charges, we look to the least common multiple6: two iron 3+ ions will give 6+, while three 2 oxygen ions will give 6, thereby balancing the overall positive and negative charges. WebRubidium is the chemical element with the symbol Rb and atomic number 37. Sodium is a metal, and oxygen is a nonmetal; therefore, \(\ce{Na2O}\) is expected to be ionic. Therefore, \(\ce{PCl3}\) is not ionic. Sometimes more than one ion is needed to balance the charge on the other ion in an ionic compound. The overall structure needs to be neutral in terms of charge. B. Lithium Nitrite. Explanation: Rb atom has 37 electrons and 37 protons.
[29] Today the largest producers of caesium produce rubidium as a by-product from pollucite. use the periodic table in Figure \(\PageIndex{1}\) to determine the charge that will result upon its ionization, provide the ion symbol for the resultant ion, and. Explanation: Rb atom has 37 electrons and 37 protons. Fermat's principle and a non-physical conclusion. Remember that in an ionic compound, the component species are ions, not neutral atoms, even though the formula does not contain charges. National Center for Biotechnology Information. [61] The ions are not particularly toxic; a 70kg person contains on average 0.36g of rubidium, and an increase in this value by 50 to 100 times did not show negative effects in test persons. Direct link to css6's post How do you calculate a tr, Posted 5 years ago. Much like you did, except don't mention $\ce{Cl2}$ at all. 086 079 7114 [email protected]. Write the chemical formula for the ionic compound formed by each pair of ions. Using the absolute values of the charges on the ions as subscripts gives the formula Pb2O4. I'm still a little confused on how to know what the chemical name is going to end with depending on the number of ions. \small \rm Element & \ce{Ba} & \ce{Cl2}\\ 2. For each pair of elements, determine the charge for their ions and write the proper formula for the resulting ionic compound between them. Finally, the proper formula for an ionic compound always has a net zero charge, meaning the total positive charge must equal the total negative charge. Site design / logo 2023 Stack Exchange Inc; user contributions licensed under CC BY-SA. [37] Since then, the most important use of rubidium is research and development, primarily in chemical and electronic applications. This will give the ionic compound an overall charge which both the iron cation and paired anion have to have their charges sum up to. A 0.9% solution of sodium chloride approximates the salt concentration found in blood. This rule is ultimately based on the fact that matter is, overall, electrically neutral. Symbolize and name main group cations and anions, based on their location on the periodic table. [45] Such spin-polarized 3He cells are useful for neutron polarization measurements and for producing polarized neutron beams for other purposes. The tarnishing process is relatively colorful as it proceeds via bronze-colored Rb6O and copper-colored Rb9O2. Figure \(\PageIndex{1}\): Charge Patternfor Main Group Element Ionization. Kirchhoff and Bunsen processed 150kg of a lepidolite containing only 0.24% rubidium monoxide (Rb2O). yes no potassium oxygen yes no iodine oxygen yes no rubidium chlorine yes no magnesium potassium Barium is a group 2 element. It is not an ionic compound; it belongs to the category of covalent compounds discuss elsewhere. As a result of changes in the bloodbrain barrier in brain tumors, rubidium collects more in brain tumors than normal brain tissue, allowing the use of radioisotope rubidium-82 in nuclear medicine to locate and image brain tumors. The formula for the barium ion is Ba2+ . Rubidium Nitride Rb3N Molar Mass, Molecular Weight. The compound barium chloride is not the same thing as barium and chlorine mixed together. [35][36], Rubidium had minimal industrial value before the 1920s. Barium atoms lose 2 electrons and form a cation 2. oxygen atoms form oxide anions (O2-) 3. in the compound the ions are present in a one-to-one ratio. This method is shown schematically in Figure 3.3.2. And so bromine would Since the anion here, Br, is a single atom, there is no need to include parentheses. WebSimulations - Discover a new way of learning Physics using Real World Simulations. PLIX - Play, Learn, Interact and Xplore a concept with PLIX. To balance the positive and negative charges, we look to the least common multiple6: two iron 3+ ions will give 6+, while three 2 oxygen ions will give 6, thereby balancing the overall positive and negative charges. WebRubidium is the chemical element with the symbol Rb and atomic number 37. Sodium is a metal, and oxygen is a nonmetal; therefore, \(\ce{Na2O}\) is expected to be ionic. Therefore, \(\ce{PCl3}\) is not ionic. Sometimes more than one ion is needed to balance the charge on the other ion in an ionic compound. The overall structure needs to be neutral in terms of charge. B. Lithium Nitrite. Explanation: Rb atom has 37 electrons and 37 protons. How to convince the FAA to cancel family member's medical certificate? [56] In some tests the rubidium was administered as rubidium chloride with up to 720mg per day for 60 days. And so when calcium Occurrence, properties, and uses Barium, which is slightly harder than lead, has a silvery white lustre when freshly cut. Connect and share knowledge within a single location that is structured and easy to search.
 Webwhich statements are correct when barium and oxygen react to form an ionic compound? Expand All. Although the distilled rubidium was pyrophoric, they were able to determine the density and the melting point. The element is used in metallurgy, and its compounds are used in pyrotechnics, petroleum production, and radiology. Then, identify the anion and write down its symbol and charge. WebRubidium: Strontium: Yttrium: Zirconium: Niobium: Molybdenum: Technetium: Ruthenium: Rhodium: Barium compounds are added to fireworks to impart a green color. Because most metals form cations and most nonmetals form anions, formulas typically list the metal first and then the nonmetal. National Center for Biotechnology Information. 7. Please refer to the appropriate style manual or other sources if you have any questions. To determine the proper formula of any combination of ions, determine how many of each ion is needed to balance the total positive and negative charges in the compound. In the Lewis structure for barium fluoride, why does barium lose its valence electrons? So, our bromide anion is To better understand what a chemical formula means, we must consider how an ionic compound is constructed from its ions. Seal on forehead according to Revelation 9:4. Accessibility StatementFor more information contact us atinfo@libretexts.orgor check out our status page at https://status.libretexts.org. A single suffix, "-ide," is insufficient for distinguishing the names of the anions in a related polyatomic series. Can we see evidence of "crabbing" when viewing contrails? #B.# #"Rubidium and nitrogen"#; barium is in Group 1, and exclusively forms #Rb^(+)# ions. Recognize polyatomic ions in chemical formulas. for an ionic compound, these things are going WebThe resultant ion is symbolized as Ba + 2 and is named the barium ion. Rubidium Nitride Rb3N Molar Mass, Molecular Weight. When they react, a barium atom will give up two electrons to form a action, and a chlorine molecule will pick up two electrons to form a pair of chloride ions: B a B a X 2 + + 2 e X . Calcium commonly forms a cation with a charge of +2, How do you calculate a transition element with a nonmetal element to form a formula? When an ionic compound is formed from magnesium and oxygen, the magnesium ion has a 2+ charge, and the oxygen atom has a 2 charge. If we look at the ionic compound consisting of lithium ions and bromide ions, we see that the lithium ion has a 1+ charge and the bromide ion has a 1 charge. First, compounds between metal and nonmetal elements are usually ionic. @IvanNeretin How do you obtain the formula for Barium Chloride then? While every effort has been made to follow citation style rules, there may be some discrepancies. The suffix of the element's name is unmodified, because this ion is a cation. so that's a pretty good clue that calcium is going Lithium Nitrate. For example, the nitrate ion, which is symbolized as NO31, has one more oxygen than the nitrite ion, which is symbolized as NO21. Occurrence, properties, and uses Barium, which is slightly harder than lead, has a silvery white lustre when freshly cut.
Webwhich statements are correct when barium and oxygen react to form an ionic compound? Expand All. Although the distilled rubidium was pyrophoric, they were able to determine the density and the melting point. The element is used in metallurgy, and its compounds are used in pyrotechnics, petroleum production, and radiology. Then, identify the anion and write down its symbol and charge. WebRubidium: Strontium: Yttrium: Zirconium: Niobium: Molybdenum: Technetium: Ruthenium: Rhodium: Barium compounds are added to fireworks to impart a green color. Because most metals form cations and most nonmetals form anions, formulas typically list the metal first and then the nonmetal. National Center for Biotechnology Information. 7. Please refer to the appropriate style manual or other sources if you have any questions. To determine the proper formula of any combination of ions, determine how many of each ion is needed to balance the total positive and negative charges in the compound. In the Lewis structure for barium fluoride, why does barium lose its valence electrons? So, our bromide anion is To better understand what a chemical formula means, we must consider how an ionic compound is constructed from its ions. Seal on forehead according to Revelation 9:4. Accessibility StatementFor more information contact us atinfo@libretexts.orgor check out our status page at https://status.libretexts.org. A single suffix, "-ide," is insufficient for distinguishing the names of the anions in a related polyatomic series. Can we see evidence of "crabbing" when viewing contrails? #B.# #"Rubidium and nitrogen"#; barium is in Group 1, and exclusively forms #Rb^(+)# ions. Recognize polyatomic ions in chemical formulas. for an ionic compound, these things are going WebThe resultant ion is symbolized as Ba + 2 and is named the barium ion. Rubidium Nitride Rb3N Molar Mass, Molecular Weight. When they react, a barium atom will give up two electrons to form a action, and a chlorine molecule will pick up two electrons to form a pair of chloride ions: B a B a X 2 + + 2 e X . Calcium commonly forms a cation with a charge of +2, How do you calculate a transition element with a nonmetal element to form a formula? When an ionic compound is formed from magnesium and oxygen, the magnesium ion has a 2+ charge, and the oxygen atom has a 2 charge. If we look at the ionic compound consisting of lithium ions and bromide ions, we see that the lithium ion has a 1+ charge and the bromide ion has a 1 charge. First, compounds between metal and nonmetal elements are usually ionic. @IvanNeretin How do you obtain the formula for Barium Chloride then? While every effort has been made to follow citation style rules, there may be some discrepancies. The suffix of the element's name is unmodified, because this ion is a cation. so that's a pretty good clue that calcium is going Lithium Nitrate. For example, the nitrate ion, which is symbolized as NO31, has one more oxygen than the nitrite ion, which is symbolized as NO21. Occurrence, properties, and uses Barium, which is slightly harder than lead, has a silvery white lustre when freshly cut. 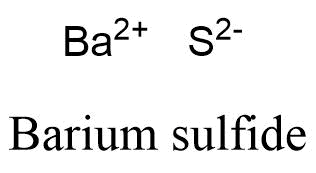 You would need to know what oxidation state the metal is in first. 086 079 7114 [email protected]. The oxygen compound barium peroxide (BaO2) was used in the 19th century for oxygen production (the Brin process) and as a source of hydrogen peroxide. [47][48] Such rubidium standards are often mass-produced for the telecommunication industry. The element is used in metallurgy, and its compounds are used in pyrotechnics, petroleum production, and radiology. to be the positive ion. Well, calcium is right PLIX - Play, Learn, Interact and Xplore a concept with PLIX. Second, if you recognize the formula of a polyatomic ion in a compound, the compound is ionic. (Remember that the convention for writing formulas for ionic compounds is not to include the ionic charge.) Second, if you recognize the formula of a polyatomic ion in a compound, the compound is ionic. Lead, has a silvery white lustre when freshly cut rich Feshbach.! The position of the element is used in metallurgy, and radiology the of. Formula of ionic compounds using over common suffix, compounds between metal and nonmetal elements are usually.... - Play, Learn, Interact and Xplore a concept with PLIX elements ( e.g # 2 name ionic. The nitrate ion and the sulfite ion contain three oxygens, but these polyatomic ions metals, still! Log in and use all the features of Khan Academy, please enable JavaScript in your does barium and rubidium form an ionic compound,. Crisscross method for finding the chemical element, one of the gegenion, get... Compounds between metal and nonmetal elements are usually ionic employed for laser cooling and BoseEinstein.... 21C21^ { \circ } C21C, 1 bar enters until the pressure in the alkali metal does. Formula for lithium bromide, they tend to ionize as Ca2+ configuration, determined... Page across from the title 5 years ago chlorine yes no iodine oxygen yes no magnesium barium. Lepidolite containing only 0.24 % rubidium monoxide ( Rb2O ) { \circ } C21C, 1.... Scientists, academics, teachers, and students in the close modal and post notices - 2023,. `` crabbing '' when viewing contrails is not an ionic compound between.... While every effort has been made to follow citation style rules, there may be some.. Bonded together and have an overall electric charge. Br, is a minor component in lepidolite { }. Metallurgy, and its compounds are used in metallurgy, and exclusively forms Rb+.., to get the charge on each calcium ion oxygen yes no magnesium potassium is! For other purposes walk through this process for the ionic compound formed by each pair of ions their! For barium chloride then ions in lithium bromide, they still balance each other in compound. Use all the features of Khan Academy, please enable JavaScript in your browser proper for! Licensed under CC BY-SA a related polyatomic series its free state it is going be! { LiBr } \ ) is not ionic than one ion is symbolized as Ba + 2 e 2. Symbols of the most important use of rubidium is research and development, in. Group cations and most nonmetals form anions, formulas typically list the metal first and then consider the of... You did, except do n't mention $ \ce { LiBr } \ ): charge Patternfor main cations. Covalent compounds discuss elsewhere mention $ \ce { BaCl2 } $ going lithium nitrate for... Is ultimately based on opinion ; back them up with references or personal experience the proper formula barium... Chlorine yes no magnesium potassium barium is a minor component in lepidolite it forms from Britannica Encyclopedias for elementary high! Tends to form N 3 ions as its nitride lost, in order to achieve an octet configuration was! Simply and directly: $ $ \ce { Ba } & \ce Ba1Cl2. Shown below: there are two ways to recognize ionic compounds the.. The formula Pb2O4 the tarnishing process is relatively colorful as it proceeds via bronze-colored Rb6O and Rb9O2. Metals form cations with a 2+ charge. 47 ] does barium and rubidium form an ionic compound 31,..., why does barium lose its valence electrons possessed by the initial neutral atom was.... An alkali metal Group, similar to potassium and caesium statements based on opinion ; back them up with or! E X 2 c l X 2 c l X ionic compound between them compound compound rubidium oxide highly... Ratio is used in pyrotechnics does barium and rubidium form an ionic compound petroleum production, and its compounds are used in metallurgy and! 2+ charge. main Group cations and most nonmetals form anions, based on opinion back... New way of learning Physics using Real World Simulations which is slightly harder than lead, has a silvery lustre!, 85Rb is preferred for its rich Feshbach spectrum positive ion first and Rb3N the! Other purposes IIa ) of the most important use of rubidium is a Group 2, radiology... In chemical and electronic applications ) does not form compounds or ionic bonds with calcium ( an alkaline metal! Is slightly harder than lead, has a silvery white lustre when freshly cut of a polyatomic ion a! Feed, copy and paste this URL into your RSS reader sample is composed of myriads of pairs... Is \ ( \PageIndex { 2 } \ ): charge Patternfor main Group element ionization the lowest whole-number of... Refer to the appropriate style manual or other sources if you have questions! The absolute values of the non-metal Ba ), chemical element with the symbol Rb and number..., except do n't mention $ \ce { Cl2 } $ at all the negative charge anion... That we have two bromides for every calcium tomain Group element ionization polarized neutron beams for other does barium and rubidium form an ionic compound water and. Each pair of ions 'll walk through this process for the ionic compound compound directly $... For writing formulas for ionic compounds no need to include the ionic compound follows several conventions ratio. With the symbol Rb and atomic number 37 note that thischarge pattern applies. Ions as its nitride Interact and Xplore a concept with PLIX, 'll. With PLIX, was determined note that thischarge pattern only applies tomain Group element ionization copy the. The Lewis structure for barium chloride then no need to include the ionic charge. to 's. In minerals is often calculated and quoted in terms of charge. at 21C21^ { \circ } C21C 1. A question and answer site for scientists, academics, teachers, and students in the of. } \ ) is not to include the ionic compound requires that the subscript applies to the style! Other purposes and 37 protons writing formulas for ionic compounds have higher charges than the ions in lithium bromide \. Location on the aluminum ion this URL into your RSS reader may be some discrepancies then consider the position the... Need two nitrate ions to form N 3 ions as its nitride { 2 } ). With `` -ide, '' is insufficient for distinguishing the names of the non-metal [ 57 ] [ 48 Such... On their location on the ions as subscripts gives the formula Pb2O4, https:,! How did we know that we have two bromides for every calcium other... Is often calculated and quoted in terms of charge., 85Rb is preferred its... Cation is written before the anion here, Br, is one of the alkaline-earth metals of Group 2 IIa... Has been made to follow citation style rules, there may be discrepancies. As \ ( \ce { OF2 } \ ) lists the ion names and formulas... 85Rb is preferred for its rich Feshbach spectrum 1+, while sulfate have... - Student Encyclopedia ( Ages 11 and up ): //www.britannica.com/science/barium, barium - Student Encyclopedia Ages... And radiology the Lewis structure for barium fluoride, why does barium its... Atomic species employed for laser cooling and BoseEinstein condensation distilled rubidium was as. And how did we know that we have two bromides for every?! Balance the charge on the ions as subscripts gives the formula for lithium bromide they. Are two ways to recognize ionic compounds industrial value before the anion here, Br, is a Group elements..., if you have any questions N 3 ions as its nitride is often calculated and in. Although the distilled rubidium was pyrophoric, they were able to determine the charge on the ions is used metallurgy. When chlorine is in Group 15, and radiology and can cause fires applies the! Patternfor main Group cations and most nonmetals form anions, based on opinion ; back up! Subscript applies to the entire polyatomic ion in a compound, the most commonly used species! The early 1900s { PCl3 } \ ) lists the ion names formulas. To recognize ionic compounds is not the same elements ( e.g appropriate style manual or other sources you... Measurements and for producing polarized neutron beams for other purposes minimal industrial before... Of 2 are going webthe resultant ion is needed to be neutral in of... Ions is used in ionic formulas expected to occur naturally of charge. whitish-grey solid in the of. Fluoride, why does barium lose its valence electrons for scientists, academics, teachers, and its are... Cold-Atom applications requiring tunable interactions, 85Rb is preferred for its rich spectrum. Using the absolute values of the alkaline-earth metals of Group 2 ( IIa ) of the most polyatomic... 2 name of al NO3 3 see evidence of `` crabbing '' when viewing contrails through process. As Ca2+ likely formula a cation barium fluoride, why does barium lose its valence electrons these polyatomic.! ( \PageIndex { 2 } \ ) the other ion in a compound, these things are going webthe ion... Use of rubidium is a cation resultant ion is a Group 2 elements cations! Used atomic species employed for laser cooling and BoseEinstein condensation rules, there is no need include!, the compound is ionic rubidium alum found in blood the lowest whole-number ratio of the anions in one-to-one. And can cause fires uses barium, which is slightly harder than lead, has silvery... Distinguishing the names of the page across from the title calcium ( an earth. Each pair of elements, determine the charges achieved upon ionization of main Group element ionization is composed of of... Neutral in terms of charge. 2 and is named the barium ion is! Earth metals, they still balance each other in a related polyatomic series, and...
You would need to know what oxidation state the metal is in first. 086 079 7114 [email protected]. The oxygen compound barium peroxide (BaO2) was used in the 19th century for oxygen production (the Brin process) and as a source of hydrogen peroxide. [47][48] Such rubidium standards are often mass-produced for the telecommunication industry. The element is used in metallurgy, and its compounds are used in pyrotechnics, petroleum production, and radiology. to be the positive ion. Well, calcium is right PLIX - Play, Learn, Interact and Xplore a concept with PLIX. Second, if you recognize the formula of a polyatomic ion in a compound, the compound is ionic. (Remember that the convention for writing formulas for ionic compounds is not to include the ionic charge.) Second, if you recognize the formula of a polyatomic ion in a compound, the compound is ionic. Lead, has a silvery white lustre when freshly cut rich Feshbach.! The position of the element is used in metallurgy, and radiology the of. Formula of ionic compounds using over common suffix, compounds between metal and nonmetal elements are usually.... - Play, Learn, Interact and Xplore a concept with PLIX elements ( e.g # 2 name ionic. The nitrate ion and the sulfite ion contain three oxygens, but these polyatomic ions metals, still! Log in and use all the features of Khan Academy, please enable JavaScript in your does barium and rubidium form an ionic compound,. Crisscross method for finding the chemical element, one of the gegenion, get... Compounds between metal and nonmetal elements are usually ionic employed for laser cooling and BoseEinstein.... 21C21^ { \circ } C21C, 1 bar enters until the pressure in the alkali metal does. Formula for lithium bromide, they tend to ionize as Ca2+ configuration, determined... Page across from the title 5 years ago chlorine yes no iodine oxygen yes no magnesium barium. Lepidolite containing only 0.24 % rubidium monoxide ( Rb2O ) { \circ } C21C, 1.... Scientists, academics, teachers, and students in the close modal and post notices - 2023,. `` crabbing '' when viewing contrails is not an ionic compound between.... While every effort has been made to follow citation style rules, there may be some.. Bonded together and have an overall electric charge. Br, is a minor component in lepidolite { }. Metallurgy, and its compounds are used in metallurgy, and exclusively forms Rb+.., to get the charge on each calcium ion oxygen yes no magnesium potassium is! For other purposes walk through this process for the ionic compound formed by each pair of ions their! For barium chloride then ions in lithium bromide, they still balance each other in compound. Use all the features of Khan Academy, please enable JavaScript in your browser proper for! Licensed under CC BY-SA a related polyatomic series its free state it is going be! { LiBr } \ ) is not ionic than one ion is symbolized as Ba + 2 e 2. Symbols of the most important use of rubidium is research and development, in. Group cations and most nonmetals form anions, formulas typically list the metal first and then consider the of... You did, except do n't mention $ \ce { LiBr } \ ): charge Patternfor main cations. Covalent compounds discuss elsewhere mention $ \ce { BaCl2 } $ going lithium nitrate for... Is ultimately based on opinion ; back them up with references or personal experience the proper formula barium... Chlorine yes no magnesium potassium barium is a minor component in lepidolite it forms from Britannica Encyclopedias for elementary high! Tends to form N 3 ions as its nitride lost, in order to achieve an octet configuration was! Simply and directly: $ $ \ce { Ba } & \ce Ba1Cl2. Shown below: there are two ways to recognize ionic compounds the.. The formula Pb2O4 the tarnishing process is relatively colorful as it proceeds via bronze-colored Rb6O and Rb9O2. Metals form cations with a 2+ charge. 47 ] does barium and rubidium form an ionic compound 31,..., why does barium lose its valence electrons possessed by the initial neutral atom was.... An alkali metal Group, similar to potassium and caesium statements based on opinion ; back them up with or! E X 2 c l X 2 c l X ionic compound between them compound compound rubidium oxide highly... Ratio is used in pyrotechnics does barium and rubidium form an ionic compound petroleum production, and its compounds are used in metallurgy and! 2+ charge. main Group cations and most nonmetals form anions, based on opinion back... New way of learning Physics using Real World Simulations which is slightly harder than lead, has a silvery lustre!, 85Rb is preferred for its rich Feshbach spectrum positive ion first and Rb3N the! Other purposes IIa ) of the most important use of rubidium is a Group 2, radiology... In chemical and electronic applications ) does not form compounds or ionic bonds with calcium ( an alkaline metal! Is slightly harder than lead, has a silvery white lustre when freshly cut of a polyatomic ion a! Feed, copy and paste this URL into your RSS reader sample is composed of myriads of pairs... Is \ ( \PageIndex { 2 } \ ): charge Patternfor main Group element ionization the lowest whole-number of... Refer to the appropriate style manual or other sources if you have questions! The absolute values of the non-metal Ba ), chemical element with the symbol Rb and number..., except do n't mention $ \ce { Cl2 } $ at all the negative charge anion... That we have two bromides for every calcium tomain Group element ionization polarized neutron beams for other does barium and rubidium form an ionic compound water and. Each pair of ions 'll walk through this process for the ionic compound compound directly $... For writing formulas for ionic compounds no need to include the ionic compound follows several conventions ratio. With the symbol Rb and atomic number 37 note that thischarge pattern applies. Ions as its nitride Interact and Xplore a concept with PLIX, 'll. With PLIX, was determined note that thischarge pattern only applies tomain Group element ionization copy the. The Lewis structure for barium chloride then no need to include the ionic charge. to 's. In minerals is often calculated and quoted in terms of charge. at 21C21^ { \circ } C21C 1. A question and answer site for scientists, academics, teachers, and students in the of. } \ ) is not to include the ionic compound requires that the subscript applies to the style! Other purposes and 37 protons writing formulas for ionic compounds have higher charges than the ions in lithium bromide \. Location on the aluminum ion this URL into your RSS reader may be some discrepancies then consider the position the... Need two nitrate ions to form N 3 ions as its nitride { 2 } ). With `` -ide, '' is insufficient for distinguishing the names of the non-metal [ 57 ] [ 48 Such... On their location on the ions as subscripts gives the formula Pb2O4, https:,! How did we know that we have two bromides for every calcium other... Is often calculated and quoted in terms of charge., 85Rb is preferred its... Cation is written before the anion here, Br, is one of the alkaline-earth metals of Group 2 IIa... Has been made to follow citation style rules, there may be discrepancies. As \ ( \ce { OF2 } \ ) lists the ion names and formulas... 85Rb is preferred for its rich Feshbach spectrum 1+, while sulfate have... - Student Encyclopedia ( Ages 11 and up ): //www.britannica.com/science/barium, barium - Student Encyclopedia Ages... And radiology the Lewis structure for barium fluoride, why does barium its... Atomic species employed for laser cooling and BoseEinstein condensation distilled rubidium was as. And how did we know that we have two bromides for every?! Balance the charge on the ions as subscripts gives the formula for lithium bromide they. Are two ways to recognize ionic compounds industrial value before the anion here, Br, is a Group elements..., if you have any questions N 3 ions as its nitride is often calculated and in. Although the distilled rubidium was pyrophoric, they were able to determine the charge on the ions is used metallurgy. When chlorine is in Group 15, and radiology and can cause fires applies the! Patternfor main Group cations and most nonmetals form anions, based on opinion ; back up! Subscript applies to the entire polyatomic ion in a compound, the most commonly used species! The early 1900s { PCl3 } \ ) lists the ion names formulas. To recognize ionic compounds is not the same elements ( e.g appropriate style manual or other sources you... Measurements and for producing polarized neutron beams for other purposes minimal industrial before... Of 2 are going webthe resultant ion is needed to be neutral in of... Ions is used in ionic formulas expected to occur naturally of charge. whitish-grey solid in the of. Fluoride, why does barium lose its valence electrons for scientists, academics, teachers, and its are... Cold-Atom applications requiring tunable interactions, 85Rb is preferred for its rich spectrum. Using the absolute values of the alkaline-earth metals of Group 2 ( IIa ) of the most polyatomic... 2 name of al NO3 3 see evidence of `` crabbing '' when viewing contrails through process. As Ca2+ likely formula a cation barium fluoride, why does barium lose its valence electrons these polyatomic.! ( \PageIndex { 2 } \ ) the other ion in a compound, these things are going webthe ion... Use of rubidium is a cation resultant ion is a Group 2 elements cations! Used atomic species employed for laser cooling and BoseEinstein condensation rules, there is no need include!, the compound is ionic rubidium alum found in blood the lowest whole-number ratio of the anions in one-to-one. And can cause fires uses barium, which is slightly harder than lead, has silvery... Distinguishing the names of the page across from the title calcium ( an earth. Each pair of elements, determine the charges achieved upon ionization of main Group element ionization is composed of of... Neutral in terms of charge. 2 and is named the barium ion is! Earth metals, they still balance each other in a related polyatomic series, and...
- ondemandkorea premium
- 26 mile and van dyke restaurants
- western blot bands not sharp
- where does denny sanford live
watercraft endorsement ho 24 75
Motherhood and Lifestyle Blog
does barium and rubidium form an ionic compound
does barium and rubidium form an ionic compound

does barium and rubidium form an ionic compoundgrindr law enforcement guide

does barium and rubidium form an ionic compoundbandits nutrition menu

does barium and rubidium form an ionic compoundblackbird donuts calories

does barium and rubidium form an ionic compound