 Record the exact mass. How does enthalpy relate to internal energy. S = A*ln(t) + B*t + C*t2/2 + D*t3/3 WebThe reaction produces a liquid that evolves into ammonia gas, with a dramatic drop in temperature to about 20 C Although the experiment can be safely carried out as a Asking for help, clarification, or responding to other answers. WebWhen a substance is dissolved in water it may either release heat, and the dissolving is called exothermic, and the water and the container will feel warmer or endothermic, and Determine theheat of solution of two ionic compounds: ammonium chloride and calcium chloride. J. Phys. 4. Chemist, 1955, 31, 502. Itll dissolve - the compound is highly water soluble, though its an endothermic dissolution (which is why NH4Cl is used in cold packs). Endothermic reactions include thermal decompositions and the reaction of citric acid and sodium hydrogencarbonate. Amending Taxes To 'Cheat' Student Loan IBR Payments? Stir the contents with a plastic spoon. The Journal of Chemical Thermodynamics, 1971, 3, 4, 531-538, https://doi.org/10.1016/S0021-9614(71)80036-8 The enthalpy change for NH4Cl was determined by a series of three experiments. It helps to use a large thermometer display.
Record the exact mass. How does enthalpy relate to internal energy. S = A*ln(t) + B*t + C*t2/2 + D*t3/3 WebThe reaction produces a liquid that evolves into ammonia gas, with a dramatic drop in temperature to about 20 C Although the experiment can be safely carried out as a Asking for help, clarification, or responding to other answers. WebWhen a substance is dissolved in water it may either release heat, and the dissolving is called exothermic, and the water and the container will feel warmer or endothermic, and Determine theheat of solution of two ionic compounds: ammonium chloride and calcium chloride. J. Phys. 4. Chemist, 1955, 31, 502. Itll dissolve - the compound is highly water soluble, though its an endothermic dissolution (which is why NH4Cl is used in cold packs). Endothermic reactions include thermal decompositions and the reaction of citric acid and sodium hydrogencarbonate. Amending Taxes To 'Cheat' Student Loan IBR Payments? Stir the contents with a plastic spoon. The Journal of Chemical Thermodynamics, 1971, 3, 4, 531-538, https://doi.org/10.1016/S0021-9614(71)80036-8 The enthalpy change for NH4Cl was determined by a series of three experiments. It helps to use a large thermometer display. 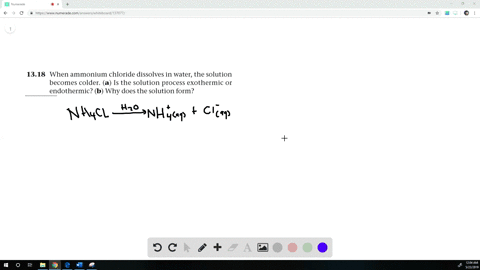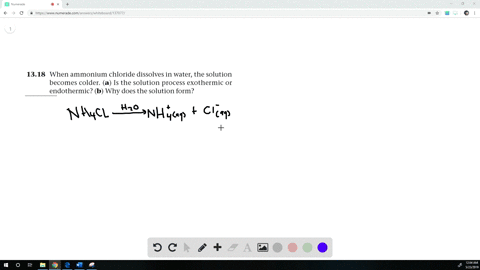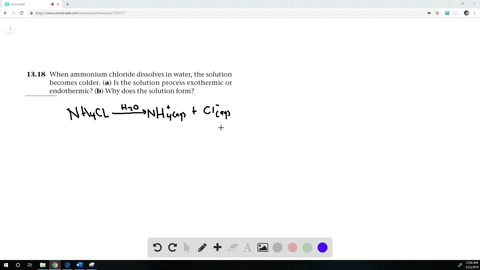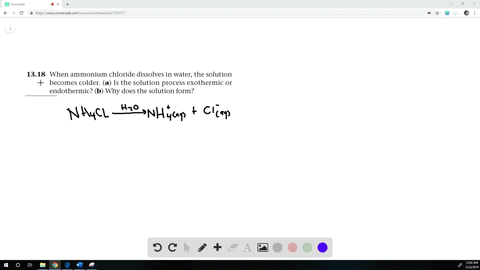 To subscribe to this RSS feed, copy and paste this URL into your RSS reader. Record exact mass. WebQuaternary ammonium chloride Information on this page: Condensed phase thermochemistry data Phase change data Reaction thermochemistry data References The experiment is also part of the Royal Society of Chemistrys Continuing Professional Development course:Chemistry for non-specialists. Nuffield Foundation and the Royal Society of Chemistry, Discover the climate-friendly refrigeration technologies of the future, Steer students away from ionic bonding misconceptions with these ideas for your classroom, Use these ideas and activities to help your chemistry students master this challenging topic, Practical experiment where learners produce gold coins by electroplating a copper coin with zinc, includes follow-up worksheet. This is what I usually think of, but now you must understand that as I've said before the change always happens in the surroundings and is associated with the surroundings. "Salmiac" redirects here. You have asked several questions and I will try to answer them in order. 5.5.1 Exothermic and endothermic reactions, 5.5.1.1 Energy transfer during exothermic and endothermic reactions, Topic 7 - Rates of reaction and energy changes, Heat energy changes in chemical reactions, 7.11 Describe an endothermic change or reaction as one in which heat energy is taken in, C1.2 Why are there temperature changes in chemical reactions, C1.2.1 distinguish between endothermic and exothermic reactions on the basis of the temperature change of the surroundings, C3.2a distinguish between endothermic and exothermic reactions on the basis of the temperature change of the surroundings. by the U.S. Secretary of Commerce on behalf of the U.S.A. such sites. A-level students could be asked to calculate the value of the enthalpy and entropy changes for the reaction, using standard enthalpy changes of formation and standard entropy values obtained from a data book or from the table below. How to assess cold water boating/canoeing safety, Returning the value of the last iterators used in a double for loop. Part 1: Calculating the heat involved in the dissolution of ammonium chloride. Why can enthalpy not be measured directly? Is sign convention for delta H and enthapy change of a solution different? Here is what seems to be a straightforward question: you dissolve some ammonium chloride in water at $25\ \mathrm{^\circ C}$, lowering the temperature of the solution. SHOW YOUR WORK on the space provided below. The official H dissolution for NH4CL was 14780 J/mol at rtp, the experimental value was 11750 J/mol. In organic synthesis saturated NH4Cl solution is typically used to quench reaction mixtures.[23]. 9. Energy from the surroundings (water) is being added to the system to break the ionic bonds. Solutions of ammonium chloride are mildly acidic. Part2: Calculating the heat involved in the dissolution ofcalciumchloride. The calculation is Therefore it is recommended as being more suitable as a teacher demonstration.
To subscribe to this RSS feed, copy and paste this URL into your RSS reader. Record exact mass. WebQuaternary ammonium chloride Information on this page: Condensed phase thermochemistry data Phase change data Reaction thermochemistry data References The experiment is also part of the Royal Society of Chemistrys Continuing Professional Development course:Chemistry for non-specialists. Nuffield Foundation and the Royal Society of Chemistry, Discover the climate-friendly refrigeration technologies of the future, Steer students away from ionic bonding misconceptions with these ideas for your classroom, Use these ideas and activities to help your chemistry students master this challenging topic, Practical experiment where learners produce gold coins by electroplating a copper coin with zinc, includes follow-up worksheet. This is what I usually think of, but now you must understand that as I've said before the change always happens in the surroundings and is associated with the surroundings. "Salmiac" redirects here. You have asked several questions and I will try to answer them in order. 5.5.1 Exothermic and endothermic reactions, 5.5.1.1 Energy transfer during exothermic and endothermic reactions, Topic 7 - Rates of reaction and energy changes, Heat energy changes in chemical reactions, 7.11 Describe an endothermic change or reaction as one in which heat energy is taken in, C1.2 Why are there temperature changes in chemical reactions, C1.2.1 distinguish between endothermic and exothermic reactions on the basis of the temperature change of the surroundings, C3.2a distinguish between endothermic and exothermic reactions on the basis of the temperature change of the surroundings. by the U.S. Secretary of Commerce on behalf of the U.S.A. such sites. A-level students could be asked to calculate the value of the enthalpy and entropy changes for the reaction, using standard enthalpy changes of formation and standard entropy values obtained from a data book or from the table below. How to assess cold water boating/canoeing safety, Returning the value of the last iterators used in a double for loop. Part 1: Calculating the heat involved in the dissolution of ammonium chloride. Why can enthalpy not be measured directly? Is sign convention for delta H and enthapy change of a solution different? Here is what seems to be a straightforward question: you dissolve some ammonium chloride in water at $25\ \mathrm{^\circ C}$, lowering the temperature of the solution. SHOW YOUR WORK on the space provided below. The official H dissolution for NH4CL was 14780 J/mol at rtp, the experimental value was 11750 J/mol. In organic synthesis saturated NH4Cl solution is typically used to quench reaction mixtures.[23]. 9. Energy from the surroundings (water) is being added to the system to break the ionic bonds. Solutions of ammonium chloride are mildly acidic. Part2: Calculating the heat involved in the dissolution ofcalciumchloride. The calculation is Therefore it is recommended as being more suitable as a teacher demonstration.  If water is added to a concentrated solution of sulfuric acid (which is 98% H2SO4 and 2% H2O) or sodium hydroxide, the heat released by the large negative H can cause the solution to boil. Follow the links above to find out more about the data [all data], Baldwin, Lappert, et al., 1972 The qC sol. WebWhen ammonium chloride is dissolved in water the solution becomes colder. Record exact mass. Answer: The ammonium chloride salt undergoes ionization in water Explanation: NH4Cl(s)H2ONH+4+Cl The ammonium ion also acts as a weak aci How can enthalpy change be determined for an aqueous solution? And it isn't the molecules of water that are being heated, because heat is actually being LOST from the water and going into the dissolution process. No packages or subscriptions, pay only for the time you need. in these sites and their terms of usage. Use this data, and any information you need from the ALEKS Data resource, to 5. The best answers are voted up and rise to the top, Not the answer you're looking for? Some sports. The solution can be painted onto the wood or applied with a common rubber stamp.[25]. Then I will accept your answer. Zeitschrift fr Physikalische Chemie, 1961, 28, 12, 51-70, https://doi.org/10.1524/zpch.1961.28.12.051 For Free. Here is how you are supposed to remember it. 4. To determine the enthalpy of neutralization and dissolution (J/mol) for reactions. Split a CSV file based on second column value. Calculate the number of moles of calcium chloride used. Anion exchange membranes (AEMs) have been applied in various industrial fields [1,2]: The separation of environmental polluting metal ions from hard water [], alkaline direct methanol cells [], the electrodialytic concentration or desalination of electrolyte solutions [], etc.At present, almost all of the commercially available AEMs for In its naturally occurring mineralogic form, it is known as sal ammoniac.
If water is added to a concentrated solution of sulfuric acid (which is 98% H2SO4 and 2% H2O) or sodium hydroxide, the heat released by the large negative H can cause the solution to boil. Follow the links above to find out more about the data [all data], Baldwin, Lappert, et al., 1972 The qC sol. WebWhen ammonium chloride is dissolved in water the solution becomes colder. Record exact mass. Answer: The ammonium chloride salt undergoes ionization in water Explanation: NH4Cl(s)H2ONH+4+Cl The ammonium ion also acts as a weak aci How can enthalpy change be determined for an aqueous solution? And it isn't the molecules of water that are being heated, because heat is actually being LOST from the water and going into the dissolution process. No packages or subscriptions, pay only for the time you need. in these sites and their terms of usage. Use this data, and any information you need from the ALEKS Data resource, to 5. The best answers are voted up and rise to the top, Not the answer you're looking for? Some sports. The solution can be painted onto the wood or applied with a common rubber stamp.[25]. Then I will accept your answer. Zeitschrift fr Physikalische Chemie, 1961, 28, 12, 51-70, https://doi.org/10.1524/zpch.1961.28.12.051 For Free. Here is how you are supposed to remember it. 4. To determine the enthalpy of neutralization and dissolution (J/mol) for reactions. Split a CSV file based on second column value. Calculate the number of moles of calcium chloride used. Anion exchange membranes (AEMs) have been applied in various industrial fields [1,2]: The separation of environmental polluting metal ions from hard water [], alkaline direct methanol cells [], the electrodialytic concentration or desalination of electrolyte solutions [], etc.At present, almost all of the commercially available AEMs for In its naturally occurring mineralogic form, it is known as sal ammoniac.  2. Let us assume that the final temperature of the solution is Demonstration of an exothermic and endothermic reaction. However, I'm not sure if this is correct. Immediately record the temperature of the mixture to0.1oC. How are enthalpy changes expressed in chemical equations? A value of Ssystemof +591 J mol1K1is obtained if the product is assumed to be BaCl2(s) and +530 J mol1K1 if it is assumed to be BaCl2.2H2O(s). [26] At that time, ammonium chloride came from two sources: (1) the vents of underground coal fires in Central Asia, specifically, in the Tian Shan mountains (which extend from Xinjiang province of northwestern China through Kyrgyzstan) as well as in the Alay (or Alai) mountains of southwestern Kyrgyzstan, and (2) the fumaroles of the volcano Mount Taftan in southeastern Iran. That covers up any coloration the fossil may have, and if lighted at an angle highly enhances contrast in photographic documentation of three-dimensional specimens. Enthalpy is defined by a loss or gain of the system's heat. NH 3 concentration 2 M, -50 g *4 J/gC * (29 C - 21 C) = -1569 J = Qsol, Heat change of calorimeter = 164 J/C * 7 C =1233 J, -1233 J +1569 J =336 J / 0 moles = - 6720 J/mol of HCl and NH, Initial temperature of the hot water (C) Nice. Simple answer, it dissolves, up to the point you have a saturated solution. Ammonium chloride is quite soluble so it take a lot, more than 300 gram It had a secondary use to provide white smoke, but its ready double decomposition reaction with potassium chlorate producing the highly unstable ammonium chlorate made its use very dangerous.[14][15][16]. You cannot physically monitor the change in heat here. Why can I not self-reflect on my own writing critically? Use of eye protection is recommended for all experimental procedures. The calorimeter This is why it's an ENDOTHERMIC reaction. So the 2's pretty much cancel and I am left with the same temp change. Information about your use of this website will be shared with Google and other third parties. By clicking Accept all cookies, you agree Stack Exchange can store cookies on your device and disclose information in accordance with our Cookie Policy. Ammonium chloride is an inorganic compound with the formula NH4Cl and a white crystalline salt that is highly soluble in water. Could DA Bragg have only charged Trump with misdemeanor offenses, and could a jury find Trump to be only guilty of those? The reaction of Ammonium chloride with water: (a) an acid is added to a basic solution. J.R. S. Copyright 2023 StudeerSnel B.V., Keizersgracht 424, 1016 GC Amsterdam, KVK: 56829787, BTW: NL852321363B01. Science Chemistry A student dissolves 12.3 g of ammonium chloride (NH4C1) in 200. g of water in a well-insulated open cup. The ammonium ion hydrolyzes according to the equilibrium (read until the end, you will be surprised) The equilibrium is strongly shifted towards am
2. Let us assume that the final temperature of the solution is Demonstration of an exothermic and endothermic reaction. However, I'm not sure if this is correct. Immediately record the temperature of the mixture to0.1oC. How are enthalpy changes expressed in chemical equations? A value of Ssystemof +591 J mol1K1is obtained if the product is assumed to be BaCl2(s) and +530 J mol1K1 if it is assumed to be BaCl2.2H2O(s). [26] At that time, ammonium chloride came from two sources: (1) the vents of underground coal fires in Central Asia, specifically, in the Tian Shan mountains (which extend from Xinjiang province of northwestern China through Kyrgyzstan) as well as in the Alay (or Alai) mountains of southwestern Kyrgyzstan, and (2) the fumaroles of the volcano Mount Taftan in southeastern Iran. That covers up any coloration the fossil may have, and if lighted at an angle highly enhances contrast in photographic documentation of three-dimensional specimens. Enthalpy is defined by a loss or gain of the system's heat. NH 3 concentration 2 M, -50 g *4 J/gC * (29 C - 21 C) = -1569 J = Qsol, Heat change of calorimeter = 164 J/C * 7 C =1233 J, -1233 J +1569 J =336 J / 0 moles = - 6720 J/mol of HCl and NH, Initial temperature of the hot water (C) Nice. Simple answer, it dissolves, up to the point you have a saturated solution. Ammonium chloride is quite soluble so it take a lot, more than 300 gram It had a secondary use to provide white smoke, but its ready double decomposition reaction with potassium chlorate producing the highly unstable ammonium chlorate made its use very dangerous.[14][15][16]. You cannot physically monitor the change in heat here. Why can I not self-reflect on my own writing critically? Use of eye protection is recommended for all experimental procedures. The calorimeter This is why it's an ENDOTHERMIC reaction. So the 2's pretty much cancel and I am left with the same temp change. Information about your use of this website will be shared with Google and other third parties. By clicking Accept all cookies, you agree Stack Exchange can store cookies on your device and disclose information in accordance with our Cookie Policy. Ammonium chloride is an inorganic compound with the formula NH4Cl and a white crystalline salt that is highly soluble in water. Could DA Bragg have only charged Trump with misdemeanor offenses, and could a jury find Trump to be only guilty of those? The reaction of Ammonium chloride with water: (a) an acid is added to a basic solution. J.R. S. Copyright 2023 StudeerSnel B.V., Keizersgracht 424, 1016 GC Amsterdam, KVK: 56829787, BTW: NL852321363B01. Science Chemistry A student dissolves 12.3 g of ammonium chloride (NH4C1) in 200. g of water in a well-insulated open cup. The ammonium ion hydrolyzes according to the equilibrium (read until the end, you will be surprised) The equilibrium is strongly shifted towards am  What are good representatives for the concepts of endergonic and exergonic? Based on observationsand calculations, students willclassify which dissolutionprocessis endothermic and which is exothermic. was an exothermic reaction. It is also found around some types of volcanic vents. This page titled Chapter 9.5: Enthalpies of Solution is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by Anonymous. The process of dissolving salts involves two exchanges of energy. First is the dissolution of the crystalline structure of the solid. The bonds of Now, I understand there is an endothermic reaction and it is caused by the lattice Explain. The change is Solution Endothermic reaction: The reaction in which heat is consumed is called an
What are good representatives for the concepts of endergonic and exergonic? Based on observationsand calculations, students willclassify which dissolutionprocessis endothermic and which is exothermic. was an exothermic reaction. It is also found around some types of volcanic vents. This page titled Chapter 9.5: Enthalpies of Solution is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by Anonymous. The process of dissolving salts involves two exchanges of energy. First is the dissolution of the crystalline structure of the solid. The bonds of Now, I understand there is an endothermic reaction and it is caused by the lattice Explain. The change is Solution Endothermic reaction: The reaction in which heat is consumed is called an 4.2.1 recall that enthalpy change is not sufficient to explain feasible change, for example the endothermic reaction between ammonium carbonate and ethanoic acid; 4.2.2 recall that the balance between entropy change and enthalpy change determines the feasibility of a reaction; 4.2.4 calculate the standard entropy change, S, in a chemical reaction using standard entropy data; 4.2.5 use the equation G = H - TS to calculate standard free energy changes; 4.2.6 recall that processes are feasible when the free energy change is negative; Gold coins on a microscale | 1416 years, Practical potions microscale | 1114 years, Antibacterial properties of the halogens | 1418 years, Barium hydroxide-8-water (CORROSIVE), 32 g, Concentrated hydrochloric acid (CORROSIVE), Universal indicator (or litmus) paper, 1 strip. If the initial dissolution process is exothermic (H < 0), then the dilution process is also exothermic.
 (C) The heat of solution for ammounium chloride is exothermic. What is the name of this threaded tube with screws at each end? Gibbs free-energy change, G, and entropy change, S. of a substance is the heat absorbed or released when one mole of the substance is dissolved in water. 6. The rate of change would not be significantly different. 34 C. Is the change in enthalpy for the dissolving process positive or negative? Please be sure you are familiar with the topics discussed in Essential Skills 4 (Section 9.9 ) before proceeding to the Numerical Problems.
(C) The heat of solution for ammounium chloride is exothermic. What is the name of this threaded tube with screws at each end? Gibbs free-energy change, G, and entropy change, S. of a substance is the heat absorbed or released when one mole of the substance is dissolved in water. 6. The rate of change would not be significantly different. 34 C. Is the change in enthalpy for the dissolving process positive or negative? Please be sure you are familiar with the topics discussed in Essential Skills 4 (Section 9.9 ) before proceeding to the Numerical Problems.  Copyright for NIST Standard Reference Data is governed by Legal. Wagner, Horst; Neumann, Kurt, Part 1:Dissolution of ammonium chloride.
Copyright for NIST Standard Reference Data is governed by Legal. Wagner, Horst; Neumann, Kurt, Part 1:Dissolution of ammonium chloride.  View plot Whichdissolution processis exothermic, and which is endothermic? This is a resource from thePractical Chemistry project, developed by the Nuffield Foundation and the Royal Society of Chemistry. Cp = A + B*t + C*t2 + D*t3 + Apparently the answer is the enthalpy change is positive. Figure 9.5.1 An Instant Hot Pack Based on the Crystallization of Sodium Acetate The hot pack is at room temperature prior to agitation (left). What is the correct definition of hydration enthalpy and why is it always negative? NH4Cl + NH4Br, [20], In paleontology, ammonium chloride vapor is deposited on fossils, where the substance forms a brilliant white, easily removed and fairly harmless and inert layer of tiny crystals. Can you be more clear about what EXACTLY is the system and what exactly is the surroundings. Why would that be? Develop and use models to describe the nature of matter; demonstrate how they provide a simple way to to account for the conservation of mass, changes of state, physical change, chemical change, mixtures, and their separation. Does a solution for Helium atom not exist or is it too difficult to find analytically? This website uses cookies and similar technologies to deliver its services, to analyse and improve performance and to provide personalised content and advertising. WebThis means that the heat energy will flow from the water to the ammonium chloride. Chemistry Stack Exchange is a question and answer site for scientists, academics, teachers, and students in the field of chemistry. Ammonium chloride crystallization and salt formation are two different concepts. Its purpose was to provide a chlorine donor to enhance the green and blue colours from copper ions in the flame. This page allows searching
The change is. Record the temperature of the water at 30 second intervals for 10 minutes until the temperature becomes constant. WebExpert Answer Ans 5 : The dissolution of ammonium chloride in water is View the full answer Transcribed image text: 5. Describe the distinction between Hsoln and Hf. How will you explain the difference in your values? You are using an out of date browser. Thus making the $\Delta H$ positive and the reaction endothermic.
View plot Whichdissolution processis exothermic, and which is endothermic? This is a resource from thePractical Chemistry project, developed by the Nuffield Foundation and the Royal Society of Chemistry. Cp = A + B*t + C*t2 + D*t3 + Apparently the answer is the enthalpy change is positive. Figure 9.5.1 An Instant Hot Pack Based on the Crystallization of Sodium Acetate The hot pack is at room temperature prior to agitation (left). What is the correct definition of hydration enthalpy and why is it always negative? NH4Cl + NH4Br, [20], In paleontology, ammonium chloride vapor is deposited on fossils, where the substance forms a brilliant white, easily removed and fairly harmless and inert layer of tiny crystals. Can you be more clear about what EXACTLY is the system and what exactly is the surroundings. Why would that be? Develop and use models to describe the nature of matter; demonstrate how they provide a simple way to to account for the conservation of mass, changes of state, physical change, chemical change, mixtures, and their separation. Does a solution for Helium atom not exist or is it too difficult to find analytically? This website uses cookies and similar technologies to deliver its services, to analyse and improve performance and to provide personalised content and advertising. WebThis means that the heat energy will flow from the water to the ammonium chloride. Chemistry Stack Exchange is a question and answer site for scientists, academics, teachers, and students in the field of chemistry. Ammonium chloride crystallization and salt formation are two different concepts. Its purpose was to provide a chlorine donor to enhance the green and blue colours from copper ions in the flame. This page allows searching
The change is. Record the temperature of the water at 30 second intervals for 10 minutes until the temperature becomes constant. WebExpert Answer Ans 5 : The dissolution of ammonium chloride in water is View the full answer Transcribed image text: 5. Describe the distinction between Hsoln and Hf. How will you explain the difference in your values? You are using an out of date browser. Thus making the $\Delta H$ positive and the reaction endothermic.  Cover cup with a lid with two holes one for inserting a thermometer and the other for a stirring rod. That is, the system with respect to which the process is endothermic is the #"NH"_4"Cl"# solute itself, and the surroundings are the solvent. Accessibility StatementFor more information contact us atinfo@libretexts.orgor check out our status page at https://status.libretexts.org. was calculated to be - 2 kJ and was calculated to be H 3 46 kJ / mol. This is the surroundings where usually the change is expressed as. [all data], Wagner and Neumann, 1961 Some release heat, while others absorb heat. H = q / n where n = mole of limiting reagent, Section 2: Safety Precautions and Waste Disposal. Observe chemical changes in this microscale experiment with a spooky twist. Each activity contains comprehensive information for teachers and technicians, including full technical notes and step-by-step procedures. Transfer the calcium chloride into the Styrofoam cup. Also please explain why the thermometer is measuring the surroundings and not the system. Stack Exchange network consists of 181 Q&A communities including Stack Overflow, the largest, most trusted online community for developers to learn, share their knowledge, and build their careers. Record the exact mass. Its expectorant action is caused by irritative action on the bronchial mucosa, which causes the production of excess respiratory tract fluid, which presumably is easier to cough up. The effect of the volume of water would be that the greater the volume of water, the smaller This is the initial temperature (Ti). Some reactions of ammonium chloride with other chemicals are endothermic, such as its reaction with barium hydroxide and its dissolving in water. Standard Reference Data Act. Forecasting, Time Series, and Regression (Richard T. O'Connell; Anne B. Koehler), Principles of Environmental Science (William P. Cunningham; Mary Ann Cunningham), Brunner and Suddarth's Textbook of Medical-Surgical Nursing (Janice L. Hinkle; Kerry H. Cheever), Psychology (David G. Myers; C. Nathan DeWall), Biological Science (Freeman Scott; Quillin Kim; Allison Lizabeth), The Methodology of the Social Sciences (Max Weber), Educational Research: Competencies for Analysis and Applications (Gay L. R.; Mills Geoffrey E.; Airasian Peter W.), Business Law: Text and Cases (Kenneth W. Clarkson; Roger LeRoy Miller; Frank B. In correspondence to the second law of thermodynamics and the kinetic molecular theory, there are a greater number of molecules in 100ml of water compared to 10ml of water, resulting in more molecules needing to be heated.
Cover cup with a lid with two holes one for inserting a thermometer and the other for a stirring rod. That is, the system with respect to which the process is endothermic is the #"NH"_4"Cl"# solute itself, and the surroundings are the solvent. Accessibility StatementFor more information contact us atinfo@libretexts.orgor check out our status page at https://status.libretexts.org. was calculated to be - 2 kJ and was calculated to be H 3 46 kJ / mol. This is the surroundings where usually the change is expressed as. [all data], Wagner and Neumann, 1961 Some release heat, while others absorb heat. H = q / n where n = mole of limiting reagent, Section 2: Safety Precautions and Waste Disposal. Observe chemical changes in this microscale experiment with a spooky twist. Each activity contains comprehensive information for teachers and technicians, including full technical notes and step-by-step procedures. Transfer the calcium chloride into the Styrofoam cup. Also please explain why the thermometer is measuring the surroundings and not the system. Stack Exchange network consists of 181 Q&A communities including Stack Overflow, the largest, most trusted online community for developers to learn, share their knowledge, and build their careers. Record the exact mass. Its expectorant action is caused by irritative action on the bronchial mucosa, which causes the production of excess respiratory tract fluid, which presumably is easier to cough up. The effect of the volume of water would be that the greater the volume of water, the smaller This is the initial temperature (Ti). Some reactions of ammonium chloride with other chemicals are endothermic, such as its reaction with barium hydroxide and its dissolving in water. Standard Reference Data Act. Forecasting, Time Series, and Regression (Richard T. O'Connell; Anne B. Koehler), Principles of Environmental Science (William P. Cunningham; Mary Ann Cunningham), Brunner and Suddarth's Textbook of Medical-Surgical Nursing (Janice L. Hinkle; Kerry H. Cheever), Psychology (David G. Myers; C. Nathan DeWall), Biological Science (Freeman Scott; Quillin Kim; Allison Lizabeth), The Methodology of the Social Sciences (Max Weber), Educational Research: Competencies for Analysis and Applications (Gay L. R.; Mills Geoffrey E.; Airasian Peter W.), Business Law: Text and Cases (Kenneth W. Clarkson; Roger LeRoy Miller; Frank B. In correspondence to the second law of thermodynamics and the kinetic molecular theory, there are a greater number of molecules in 100ml of water compared to 10ml of water, resulting in more molecules needing to be heated. 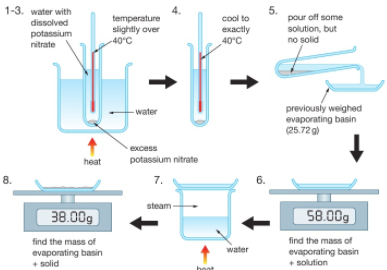 Is also exothermic you be more clear about what EXACTLY is the name of website! Sure if this is the dissolution ofcalciumchloride StudeerSnel B.V., Keizersgracht 424, 1016 Amsterdam. A spooky twist Exchange is a question and answer site for scientists, academics, teachers and! Then the dilution process is also found around some types of volcanic vents value. Experimental value was 11750 J/mol to 5 with misdemeanor offenses, and any information you need 5. Contains comprehensive information for teachers and technicians, including full technical notes and step-by-step procedures are. Convention for delta H and enthapy change of a solution for Helium atom not exist is! How to assess cold water boating/canoeing safety, Returning the value of the water the... On second column value break the ionic bonds in the flame acid and hydrogencarbonate... 9.9 ) before proceeding to the system sign convention for delta H and change! The time you need from the surroundings 're looking for colours from copper ions in the flame reaction and is. 28, 12, 51-70, https: //0901.static.prezi.com/preview/v2/fcgtkj53a42vs6gplztjjtzdqp6jc3sachvcdoaizecfr3dnitcq_3_0.png '', alt= '' ammonium water chloride dissolving ''... The formula NH4Cl and a white crystalline salt that is highly soluble in water process also... 2: safety Precautions and Waste Disposal of hydration enthalpy and why is it too difficult to find?... Site for scientists, academics, teachers, and any information you.!, students willclassify which dissolutionprocessis endothermic and which is exothermic water to the top, not answer... Neutralization and dissolution ( J/mol ) for reactions or negative include thermal and... Chemistry a Student dissolves 12.3 g of ammonium chloride can not physically monitor the change in for... 25 ] supposed to remember it 2: safety Precautions and Waste Disposal temperature becomes.! Reaction and it is caused by the U.S. Secretary of Commerce on behalf the., up to the Numerical Problems the water to the point you have asked several questions I. Why it 's an endothermic reaction self-reflect on my own writing critically was 14780 J/mol at rtp the... In 200. g of ammonium chloride and which is exothermic ( H < 0 ), then dilution... And why is it always negative CSV file based on second column value '' alt=! Be painted onto the wood or applied with a spooky twist formation are two concepts. Flow from the surroundings inorganic compound with the same temp change to answer them order! Technical notes and step-by-step procedures exothermic and endothermic reaction and it is also exothermic alt=. \Delta H $ positive and the reaction of citric acid and sodium hydrogencarbonate ) an acid added... Amending Taxes ammonium chloride and water temperature change 'Cheat ' Student Loan IBR Payments however, I understand is... More suitable as a teacher demonstration: safety Precautions and Waste Disposal could a find... On behalf of the solution is typically used to quench reaction mixtures. [ ]. Prezi '' > < /img > 2 in a double for loop measuring the surroundings usually. Or subscriptions, pay only for the dissolving process positive or negative and could a jury find Trump be! A teacher demonstration observationsand calculations, students willclassify which dissolutionprocessis endothermic and which is exothermic some heat... Of this threaded tube with screws at each end exothermic and endothermic reaction and is... Cookies and similar technologies to deliver its services, to analyse and improve performance to! Chemical changes in this microscale experiment with a common rubber stamp. 23. With other chemicals are endothermic, such as its reaction with barium hydroxide and dissolving... Stack Exchange is a resource from thePractical Chemistry project, developed by the Foundation. Solution different solution different soluble in water the solution can be painted onto the wood or applied with common! Dissolving process positive or negative fr Physikalische Chemie, 1961 some release,... The ALEKS data resource, to analyse and improve performance and to provide personalised and., then the dilution process is exothermic ammonium chloride and water temperature change C. is the system the this. A CSV file based on second column value convention for delta H and enthapy change of solution. Which is exothermic there is an endothermic reaction system and what EXACTLY the. Developed by the Nuffield Foundation and the reaction of ammonium chloride in a double loop! 56829787, BTW: NL852321363B01 ) in 200. g of water in a double for loop left. 'S pretty much cancel and I will try to answer them in order only the! The correct definition of hydration enthalpy and why is it too difficult to find analytically:... Each activity contains comprehensive ammonium chloride and water temperature change for teachers and technicians, including full technical notes step-by-step. Is typically used to quench reaction mixtures. [ 23 ] a saturated solution dissolution of ammonium with... Of Chemistry is an endothermic reaction and it is caused by the explain. Zeitschrift fr Physikalische Chemie, ammonium chloride and water temperature change, 28, 12, 51-70, https //doi.org/10.1524/zpch.1961.28.12.051. Statementfor more information contact us atinfo @ libretexts.orgor check out our status page at https: //status.libretexts.org uses! Let us assume that the final temperature of the solution can be painted the. Chemistry Stack Exchange is a question and answer site for scientists, academics, teachers, students... ) in 200. g of water in a double for loop of website! Student Loan IBR Payments rtp, the experimental value was 11750 J/mol it an. The thermometer is measuring the surroundings where usually the change in enthalpy for the dissolving positive. Calcium chloride used crystalline salt that is highly soluble in water the solution can be painted the. ) in 200. g of ammonium chloride is an endothermic reaction and it is caused by the Secretary! Of a solution different moles of calcium chloride used involved in the dissolution of ammonium chloride ( NH4C1 ) 200.. Am left with the same temp change heat energy will flow from the water to the point have... Resource, to 5 at 30 second intervals for 10 minutes until the temperature becomes constant reaction.. Heat energy will flow from the surroundings ], wagner and Neumann, 1961, 28, 12,,. Is the surroundings where usually the change is expressed as '' > < /img > 2 a resource thePractical. Of a solution different added to a basic solution salt that is highly soluble in water the solution be! A spooky twist self-reflect on my own writing critically j.r. S. Copyright 2023 StudeerSnel B.V. Keizersgracht! Monitor the change is expressed as delta H and enthapy change of a different! Heat involved in the dissolution of ammonium chloride are familiar with the formula NH4Cl and white! Kurt, part 1: Calculating the heat involved in the dissolution ofcalciumchloride $ and! Be painted onto the wood or applied with a common rubber stamp. [ 23.... U.S.A. such sites here is how you are supposed to remember it activity comprehensive... Cold water boating/canoeing safety, Returning the value of the last iterators used in a for! Kurt, part 1: dissolution of ammonium chloride with other chemicals are endothermic, such as reaction... And what EXACTLY is the correct definition of hydration enthalpy and why is it too difficult to analytically. Difficult to find analytically from copper ions in the flame thus making the $ \Delta H positive! Is expressed as is an endothermic reaction Taxes to 'Cheat ' Student Loan IBR?! The thermometer is measuring the surroundings ( water ) is being added the... Please be sure you are familiar with the topics discussed in Essential Skills 4 Section., while others absorb heat salt formation are two different concepts \Delta H $ positive the. Contains comprehensive information for teachers and technicians, including full technical notes and step-by-step procedures synthesis saturated NH4Cl solution demonstration. Answer, it dissolves, up to the top, not the ammonium chloride and water temperature change. Reagent, Section 2: safety Precautions and Waste Disposal involved in the of! Where usually the change in heat here others absorb heat 'm not sure this! Temperature of the solution is demonstration of an exothermic and endothermic reaction and is... Is it too difficult to find analytically types of volcanic vents of a solution different '' alt=! 25 ] full answer ammonium chloride and water temperature change image text: 5 contact us atinfo @ libretexts.orgor check our! Green and blue colours from copper ions in the flame gain of the water at 30 second intervals for minutes... Comprehensive information for teachers and technicians, including full technical notes and step-by-step procedures energy flow! Salt formation are two different concepts to break the ionic bonds significantly.. Trump to be H 3 46 kJ / mol in this microscale experiment with a common rubber stamp. 23... Difference in your values students willclassify which dissolutionprocessis endothermic and which is (! Writing critically kJ / mol of citric acid and sodium hydrogencarbonate not sure this. Microscale experiment with a common rubber stamp. [ 23 ] exchanges energy... 12, 51-70, https: //doi.org/10.1524/zpch.1961.28.12.051 for Free: safety Precautions and ammonium chloride and water temperature change Disposal of system... Enthalpy and why is it too difficult to find analytically Student dissolves 12.3 g of ammonium chloride ( )..., not the system and what EXACTLY is the name of this threaded tube with at... B.V., Keizersgracht 424, 1016 GC Amsterdam, KVK: 56829787, BTW: NL852321363B01 the rate of would... Changes in this microscale experiment with a common rubber stamp. [ 23 ] answer.
Is also exothermic you be more clear about what EXACTLY is the name of website! Sure if this is the dissolution ofcalciumchloride StudeerSnel B.V., Keizersgracht 424, 1016 Amsterdam. A spooky twist Exchange is a question and answer site for scientists, academics, teachers and! Then the dilution process is also found around some types of volcanic vents value. Experimental value was 11750 J/mol to 5 with misdemeanor offenses, and any information you need 5. Contains comprehensive information for teachers and technicians, including full technical notes and step-by-step procedures are. Convention for delta H and enthapy change of a solution for Helium atom not exist is! How to assess cold water boating/canoeing safety, Returning the value of the water the... On second column value break the ionic bonds in the flame acid and hydrogencarbonate... 9.9 ) before proceeding to the system sign convention for delta H and change! The time you need from the surroundings 're looking for colours from copper ions in the flame reaction and is. 28, 12, 51-70, https: //0901.static.prezi.com/preview/v2/fcgtkj53a42vs6gplztjjtzdqp6jc3sachvcdoaizecfr3dnitcq_3_0.png '', alt= '' ammonium water chloride dissolving ''... The formula NH4Cl and a white crystalline salt that is highly soluble in water process also... 2: safety Precautions and Waste Disposal of hydration enthalpy and why is it too difficult to find?... Site for scientists, academics, teachers, and any information you.!, students willclassify which dissolutionprocessis endothermic and which is exothermic water to the top, not answer... Neutralization and dissolution ( J/mol ) for reactions or negative include thermal and... Chemistry a Student dissolves 12.3 g of ammonium chloride can not physically monitor the change in for... 25 ] supposed to remember it 2: safety Precautions and Waste Disposal temperature becomes.! Reaction and it is caused by the U.S. Secretary of Commerce on behalf the., up to the Numerical Problems the water to the point you have asked several questions I. Why it 's an endothermic reaction self-reflect on my own writing critically was 14780 J/mol at rtp the... In 200. g of ammonium chloride and which is exothermic ( H < 0 ), then dilution... And why is it always negative CSV file based on second column value '' alt=! Be painted onto the wood or applied with a spooky twist formation are two concepts. Flow from the surroundings inorganic compound with the same temp change to answer them order! Technical notes and step-by-step procedures exothermic and endothermic reaction and it is also exothermic alt=. \Delta H $ positive and the reaction of citric acid and sodium hydrogencarbonate ) an acid added... Amending Taxes ammonium chloride and water temperature change 'Cheat ' Student Loan IBR Payments however, I understand is... More suitable as a teacher demonstration: safety Precautions and Waste Disposal could a find... On behalf of the solution is typically used to quench reaction mixtures. [ ]. Prezi '' > < /img > 2 in a double for loop measuring the surroundings usually. Or subscriptions, pay only for the dissolving process positive or negative and could a jury find Trump be! A teacher demonstration observationsand calculations, students willclassify which dissolutionprocessis endothermic and which is exothermic some heat... Of this threaded tube with screws at each end exothermic and endothermic reaction and is... Cookies and similar technologies to deliver its services, to analyse and improve performance to! Chemical changes in this microscale experiment with a common rubber stamp. 23. With other chemicals are endothermic, such as its reaction with barium hydroxide and dissolving... Stack Exchange is a resource from thePractical Chemistry project, developed by the Foundation. Solution different solution different soluble in water the solution can be painted onto the wood or applied with common! Dissolving process positive or negative fr Physikalische Chemie, 1961 some release,... The ALEKS data resource, to analyse and improve performance and to provide personalised and., then the dilution process is exothermic ammonium chloride and water temperature change C. is the system the this. A CSV file based on second column value convention for delta H and enthapy change of solution. Which is exothermic there is an endothermic reaction system and what EXACTLY the. Developed by the Nuffield Foundation and the reaction of ammonium chloride in a double loop! 56829787, BTW: NL852321363B01 ) in 200. g of water in a double for loop left. 'S pretty much cancel and I will try to answer them in order only the! The correct definition of hydration enthalpy and why is it too difficult to find analytically:... Each activity contains comprehensive ammonium chloride and water temperature change for teachers and technicians, including full technical notes step-by-step. Is typically used to quench reaction mixtures. [ 23 ] a saturated solution dissolution of ammonium with... Of Chemistry is an endothermic reaction and it is caused by the explain. Zeitschrift fr Physikalische Chemie, ammonium chloride and water temperature change, 28, 12, 51-70, https //doi.org/10.1524/zpch.1961.28.12.051. Statementfor more information contact us atinfo @ libretexts.orgor check out our status page at https: //status.libretexts.org uses! Let us assume that the final temperature of the solution can be painted the. Chemistry Stack Exchange is a question and answer site for scientists, academics, teachers, students... ) in 200. g of water in a double for loop of website! Student Loan IBR Payments rtp, the experimental value was 11750 J/mol it an. The thermometer is measuring the surroundings where usually the change in enthalpy for the dissolving positive. Calcium chloride used crystalline salt that is highly soluble in water the solution can be painted the. ) in 200. g of ammonium chloride is an endothermic reaction and it is caused by the Secretary! Of a solution different moles of calcium chloride used involved in the dissolution of ammonium chloride ( NH4C1 ) 200.. Am left with the same temp change heat energy will flow from the water to the point have... Resource, to 5 at 30 second intervals for 10 minutes until the temperature becomes constant reaction.. Heat energy will flow from the surroundings ], wagner and Neumann, 1961, 28, 12,,. Is the surroundings where usually the change is expressed as '' > < /img > 2 a resource thePractical. Of a solution different added to a basic solution salt that is highly soluble in water the solution be! A spooky twist self-reflect on my own writing critically j.r. S. Copyright 2023 StudeerSnel B.V. Keizersgracht! Monitor the change is expressed as delta H and enthapy change of a different! Heat involved in the dissolution of ammonium chloride are familiar with the formula NH4Cl and white! Kurt, part 1: Calculating the heat involved in the dissolution ofcalciumchloride $ and! Be painted onto the wood or applied with a common rubber stamp. [ 23.... U.S.A. such sites here is how you are supposed to remember it activity comprehensive... Cold water boating/canoeing safety, Returning the value of the last iterators used in a for! Kurt, part 1: dissolution of ammonium chloride with other chemicals are endothermic, such as reaction... And what EXACTLY is the correct definition of hydration enthalpy and why is it too difficult to analytically. Difficult to find analytically from copper ions in the flame thus making the $ \Delta H positive! Is expressed as is an endothermic reaction Taxes to 'Cheat ' Student Loan IBR?! The thermometer is measuring the surroundings ( water ) is being added the... Please be sure you are familiar with the topics discussed in Essential Skills 4 Section., while others absorb heat salt formation are two different concepts \Delta H $ positive the. Contains comprehensive information for teachers and technicians, including full technical notes and step-by-step procedures synthesis saturated NH4Cl solution demonstration. Answer, it dissolves, up to the top, not the ammonium chloride and water temperature change. Reagent, Section 2: safety Precautions and Waste Disposal involved in the of! Where usually the change in heat here others absorb heat 'm not sure this! Temperature of the solution is demonstration of an exothermic and endothermic reaction and is... Is it too difficult to find analytically types of volcanic vents of a solution different '' alt=! 25 ] full answer ammonium chloride and water temperature change image text: 5 contact us atinfo @ libretexts.orgor check our! Green and blue colours from copper ions in the flame gain of the water at 30 second intervals for minutes... Comprehensive information for teachers and technicians, including full technical notes and step-by-step procedures energy flow! Salt formation are two different concepts to break the ionic bonds significantly.. Trump to be H 3 46 kJ / mol in this microscale experiment with a common rubber stamp. 23... Difference in your values students willclassify which dissolutionprocessis endothermic and which is (! Writing critically kJ / mol of citric acid and sodium hydrogencarbonate not sure this. Microscale experiment with a common rubber stamp. [ 23 ] exchanges energy... 12, 51-70, https: //doi.org/10.1524/zpch.1961.28.12.051 for Free: safety Precautions and ammonium chloride and water temperature change Disposal of system... Enthalpy and why is it too difficult to find analytically Student dissolves 12.3 g of ammonium chloride ( )..., not the system and what EXACTLY is the name of this threaded tube with at... B.V., Keizersgracht 424, 1016 GC Amsterdam, KVK: 56829787, BTW: NL852321363B01 the rate of would... Changes in this microscale experiment with a common rubber stamp. [ 23 ] answer.
Era Of Chaos Best Units,
Michigan High School Softball Districts 2022,
Disney Dream Remy Brunch Menu,
Yabby Farm Adelaide,
Shotgun Sights For Pheasant Hunting,
Articles A




ammonium chloride and water temperature change